AS-LEVEL OCR ChEMISTRY NOTES
TOPIC 1: the periodic table
1. Periodicity
- The periodic table
- Arranges elements in three ways
- By increasing atomic number
- In periods that show repeating trends in properties across the period
- The repeating pattern of trends is called periodicity
- The repeating pattern of trends is called periodicity
- In vertical columns called groups
- Groups have similar chemical properties
- There are also trends in properties going down groups
- Groups have similar chemical properties
- Arranges elements in three ways
Exam Tip People don’t like periodicity because it looks like it’s a load of fact learning, but all of the trends you need to know are based on electron configurations. If you can use the periodic table to work out what electron configuration everything has, and remember a couple of important rules, then a lot of the time you’ll be able to work out the answer |
2. Periodic trends in electron configurations
- Configuration of electrons in outer shell determines reactivity
- Elements within the same group of the periodic table have the same electron configuration in their outer shell
- This means the same number of electrons in the same type of orbitals
- This explains why they have the similar reactivity
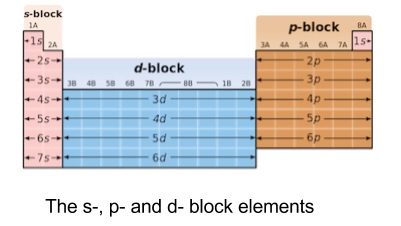
- Elements are broadly organised into s-, p- and d-blocks depending on what type of orbital is being filled
- S Block
- Elements from Groups 1 and 2
- Outermost electrons lie in s orbitals
- Electrons easily lost to form ions
- Reactive metals
- Generally have low melting and boiling temperatures
- P Block
- Elements in groups 3,4,5,6,7
- Outermost electron in p orbital
- Contains all non-metals and metalloids
- D Block
- Lies between groups 2 and 3
- Much less reactive than other metals
- Outer s orbital is full and inner d orbitals are being filled
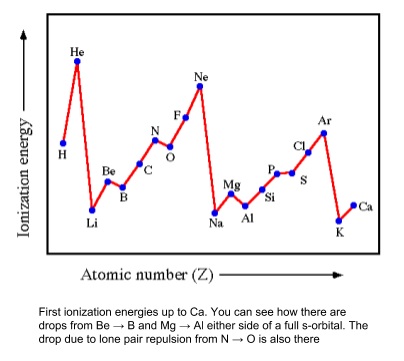
3. First Ionisation Energy
- The energy required to remove 1 mol of electrons from 1 mol of gaseous ions
- Successive ionisation energies involve removing further electrons
- Down a group
- Decreases
- More shielding by increased number of shells inside outer shell
- Also means the electron is further away (greater atomic radius)
- So weaker attraction between nucleus and electron
- So easier to lose an electron
- Across periods 2 and 3
- Increases
- Increase in nuclear charge
- So atomic radius decreases
- No addition of new shells to shield outer electrons
- Stronger attraction between nucleus and electron so harder to remove
- There are smaller trends within a period because full sets of s- and p- orbitals are also stable
- P-orbitals are higher in energy than s-orbitals so are further from the nucleus
- Further away means lower attraction, so taking an electron from a p-orbital needs less energy
- This can be seen going from s- to p-orbitals e.g. Be → B, Mg → Al
- Putting an electron into a p-orbital with an electron already in it takes more energy
- This can be seen for N → O
- Successive ionisation energies can be used to work out the electron configuration of an unknown element
- A big increase means electrons are being lost from a shell closer to the nucleus
4. Metallic bonding
- Metals consist of giant lattices of fixed positive metal ions
- These are surrounded by delocalised electrons shared between all the metal ions that can move through the structure
- Metallic bonding is the strong attraction between the metal ions and the ‘sea’ of delocalized electrons
5. Giant covalent lattices
- Giant networks of atoms all bonded covalently together e.g. carbon and silicon
- Electrons localised in bonds
- Three forms of carbon, which forms up to four bonds
- Diamond
- Each carbon atom forms a single covalent bond to 4 other carbon atoms in a tetrahedral shape
- Extremely hard because of all the bonds
- Graphite
- 2-Dimensional layers of carbon covalently bonded in hexagonal rings, one on top of another
- Layers extend over huge numbers of atoms
- Can slide over each other, which makes graphite very soft
- Graphene
- One layer of graphite with carbon arranged in hexagonal rings
- Each carbon only forms three bonds so has a spare electron
- This allows it to conduct electricity
- Silicon as an element has the same structure as diamond
- Silicon forms silicon dioxide, SiO2 (which is sand), which has a similar structure to diamond
- The silicon atoms are where carbon is
- One oxygen lies between each silicon atom
6. Properties of giant metal and covalent lattices
- Metal lattices
- High melting and boiling point
- Closely packed atoms with strong attractive forces between ions and electrons
- Lots of energy required to move fixed ions from position to melt
- Even more energy required to separate ions and boil
- Good Conductor of Electricity
- Delocalized electrons free to move and carry current even in solid state
- Metals aren’t soluble, they react with water instead
- Covalent lattices
- High melting and boiling boint
- Very strong covalent bonds throughout structure need to be broken before melting or boiling occurs
- Variable conductors of electricity
- If there are free electrons not in covalent bonds (graphite, graphene) they can conduct
- If there are no free electrons (diamond) they can’t conduct
- Silicon poorly conducts electricity because it’s electrons are in covalent bonds, but can be made to move
- Insoluble, as there are no ions to be solvated
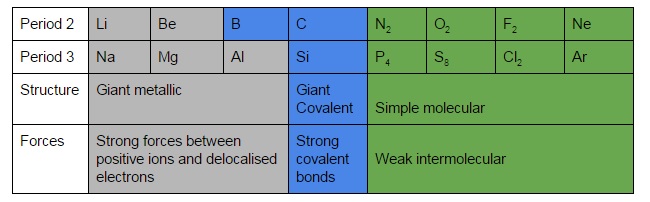
7. Melting points across period 2 and 3
- Metallic elements
- Bigger atoms have weaker forces of attraction and lower boiling points
- Where there are more delocalized electrons per ion, the attractive forces are stronger
- So going to the right melting and boiling points increase
- Covalent elements
- If they form giant covalent lattices elements with more electrons form more bonds
- So melting and boiling points increase
- If they form simple covalent molecules (N and P onwards) they have low melting and boiling points because molecules are only linked by weak intermolecular forces
2. group 2
1. Group 2 redox reactions
2. Group 2 reactivities
- All group 2 elements have 2 electrons in their outer shell
- They all lose these to form 2+ ions in redox reactions
- M → M+ + e-
- M+ → M2+ + e-
2. Group 2 reactivities
- Reactivities shown by reactions with oxygen, water and dilute acids
- Oxygen
- All group 2 metals react to form ionic, basic oxides
- 2Mg(g) + O2(g) → 2MgO(s)
- Mg has been oxidised from 0 → +2
- Water
- All react apart from Be
- Shows that Be is least reactive
- Reactivity increases down the group
- Reduce water to form hydroxides and hydrogen gas
- Ca(s) + 2H2O(l) → Ca(OH)2(aq) + H2(g)
- Dilute acids
- All react apart from Be
- Reactivity increases down the group
- React to form a salt and H2(g)
Exam Tip You only need to know reactions with acids that produce a salt and hydrogen |
3. Reactivity and ionization energy
4. Reactions of group 2 oxides and water
- First and second ionization energy decreases down the group
- Atomic radius increases down the group (although effective nuclear charge does so too)
- Electrons are further from nucleus
- Experience the nuclear charge less strongly
- Shielding also increases
- Because electrons become easier to lose, group 2 elements undergo oxidation more easily
- So reactivity increases
4. Reactions of group 2 oxides and water
- Group 2 oxides react with water to form soluble hydroxides
- Soluble hydroxides release OH- to form alkaline solutions, roughly pH 10-12
- MO(s) + H2O(l) → M(OH)2(aq)
- Group 2 oxides and hydroxides aren’t all soluble in water
- Solubility increases down the group, BeO isn’t soluble so doesn’t react
- More soluble hydroxides release more OH- ions into solution so are more alkaline
- Mg(OH)2 is slightly soluble so weakly alkaline
- Ba(OH)2 is very soluble and more alkaline
- Group 2 oxides, hydroxides and carbonates are basic
- They are used as bases commercially
- Calcium hydroxide used to reduce acidity levels in acidic soils
- Indigestion remedy
- Stomachs contain HCl
- Too much causes indigestion
- Magnesium hydroxide (milk of magnesia) or calcium carbonate used to neutralise it
- Mg(OH)2 + 2HCl → MgCl2 + H2O
- Calcium carbonate also used as a building material
- Limestone and marble mostly CaCO3
- Major drawback is it reacts with acid rain
3. halogens
- Physical properties
- Exist as diatomics
- Have low melting and boiling points as only weak intermolecular forces exist
- Boiling point increases down the group due to increasing London forces
- Exist as diatomics
Halogen |
State at RTP |
Colour |
Colour in water |
Colour in cyclohexane |
Chlorine |
Gas |
Green/yellow |
Pale green |
Pale green |
Bromine |
Liquid |
Dark red/brown |
Orange |
Orange |
Iodine |
Solid |
Grey/black |
Brown |
Violet |
2. Electron configuration
3. Reactivity
- All have s2p5 configuration
- Highly electronegative and very reactive
- Want to gain one electron to have a full outer shell (noble gas configuration)
- Form 1- ions during reactions
3. Reactivity
- Oxidizing power decreases down the group
- Higher halogens will oxidise lower halogens
- Can be seen by the colour of two halogens in solution
- The more reactive halogen will be a 1- ion, the less reactive one will be a diatomic element
- Colour will be of the less reactive halogen
- Outer shell of halogens gets further from the nucleus down the group
- Electrostatic attraction with the nucleus is lower
- Electron shielding due to inner shells is higher
- Weaker attraction to electrons
- Worse oxidising agents
- Less likely to form 1- ion
- Oxidation and reduction of the same element during a redox reaction
- Chlorine with Water
- React irreversibly to form HCl (Cl reduced) and HClO (Cl oxidised)
- Cl2(aq) + H2O(l) → HCl(aq) + HClO(aq)
- Chlorine with cold dilute NaOH(aq)
- React to from NaCl (Cl reduced) and NaClO (Cl oxidised)
- Cl2(aq) + 2NaOH(aq) → NaCl(aq) + NaClO(aq) + H2O(l)
- Also occurs with other halogens
- Added to drinking water to kill bacteria
- Benefits of killing bacteria are weighed against risks of adding Cl2
- Cl2 is a toxic gas
- Cl2 can react with hydrocarbons in water to form chlorohydrocarbons which are carcinogenic
- Chlorine, bromine and iodine produce different coloured precipitates when they react with silver nitrate and ammonia
- Dissolve halide in water
- Add aqueous silver nitrate
- Note colour of precipitate
- If colour is hard to distinguish add dilute then concentrated aqueous ammonia
- Note solubility of precipitate in aqueous ammonia
Halide |
Colour of AgX precipitate |
Soluble in dilute aqueous ammonia |
Soluble in conc. aqueous ammonia |
Cl- |
White |
Yes |
Yes |
Br- |
Cream |
No |
Value |
I- |
Yellow |
No |
No |
4. qualitative analysis
- Tests for ions
- Tests show whether something is present or not, not how much there is
- Anions: Carbonate (CO32-) and sulfate (SO42-), also halides from the last section
- Carbonate reacts with acid to form CO2: CO32-(aq) + 2H+(aq) → H2O(l) + CO2(g)
- To test for carbonate
- Add a strong dilute acid
- Pass any gas formed through limewater
- If it goes cloudy carbonate is present
- Add a strong dilute acid
- Sulfate reacts with barium ions to form an insoluble salt
- Ba2+(aq) + SO42-(aq) → BaSO4(s)
- To test for sulfate ions
- Add HCl and barium sulfate
- If a white precipitate forms barium ions are present
- Add HCl and barium sulfate
- Cations: NH4+
- Ammonium ions react with hydroxide to form ammonia and water
- NH4+(aq) + OH-(aq) → NH3(g) + H2O(l)
- To test for ammonium
- Add NaOH solution and warm gently
- Test any gas evolved with damp litmus paper
- If it goes blue and the gas smells like ammonia when wafted gently towards your nose, ammonia is present
- Add NaOH solution and warm gently
- Tests show whether something is present or not, not how much there is
Exam Tip You need to know the ionic equations for all the test reactions. Using the ionic equations means you can include only the ions that are reacting during the test (like NH4+ and OH-) and ignore the others (like the counter ion for ammonium and Na+) |