AS-LEVEL OCR ChEMISTRY NOTES
TOPIC 2: physical chemistry
1. enthalpy changes
1. Enthalpy changes in reactions
- Enthalpy, H, is the amount of thermal energy in a system in kJ
- Can’t measure H, but in some reactions there is a change of enthalpy, ΔH
- ΔH is the heat exchanged between system (atoms and bonds involved) and surroundings (everything else)
- Normally kJmol-1
- ΔH = Hproducts - Hreactants, can be positive or negative
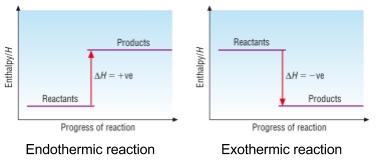
- Negative = Exothermic
- Heat flows from the system to the surroundings (energy is released)
- Energy of system has decreased
- Positive = Endothermic
- Heat flows from the surroundings into the system (energy is absorbed)
- Energy of system has increased
Exam Tip Exothermic reactions give out heat and endothermic reactions take it up, so an exothermic reaction in a test-tube will feel warm and an endothermic one will feel cold. It seems weird that an exothermic reaction should have a negative ΔH because it’s giving out heat, but it’s just the way that ΔH is defined as Hproducts - Hreactants |
2. Energy profile diagrams
- Change in enthalpy can be shown on a diagram
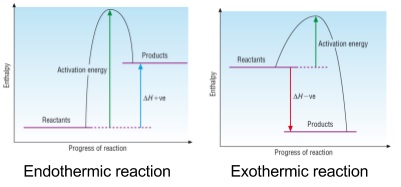
3. Activation energy, Ea
- Minimum amount of energy that the particles need to react during a reaction
- Looks like a hump on an energy profile diagram
- Even though energy comes out of an exothermic reaction still need energy to overcome the activation energy to make the reaction happen
Exam Tip You need to remember to show enthalpy change and activation energy with arrows pointing the right way. Learn the ways they’re drawn in the diagrams. Exothermic arrows go down, endothermic go up, activation energy arrows always go up and are shown going from reactants to products. |
4. Important terms
5. Determining enthalpy changes from experiment
- Standard conditions: 100kPa and 298K
- Standard states: physical state an atom or molecule is in at standard conditions
- All enthalpy changes measured in kJmol-1 and under standard conditions
- Enthalpy change of reaction, ΔrH: Energy change of a reaction
- Enthalpy change of formation, ΔfH: The enthalpy change when one mole of the compound is formed from its elements
- Enthalpy change of combustion, ΔcH: The enthalpy change that occurs when one mole of a substance is completely burned in oxygen
- Enthalpy change of neutralisation, ΔneutH: The energy change when an acid and a base react to form 1 mole of water
5. Determining enthalpy changes from experiment
- Measure enthalpy change from experiment by measuring change in temperature
- Use the relationship q = mcΔT to find enthalpy change
- q = heat exchanged with surroundings, normally in J
- m = mass of substance changing temperature, normally in g
- c = specific heat capacity, energy required to raise the temperature of a gram of substance by 1K, normally in Jg-1K-1
- ΔT = temperature change in K
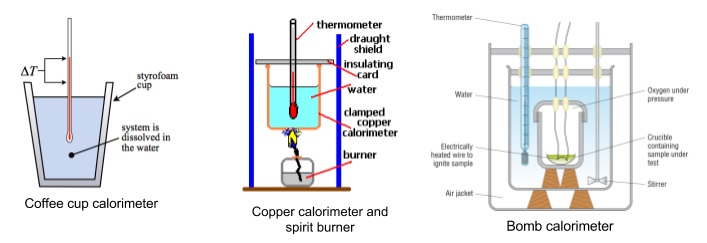
- Three experimental methods: coffee cup calorimeter, copper calorimeter and spirit burner, bomb calorimeter
- All heat up water and help minimise amount of heat lost to surroundings
- Coffee cup calorimeter
- Add one liquid to coffee cup, take temperature every minute until it stabilises (4 minutes)
- Add second liquid to cup, measure temperature every minute from 6 minutes onward
- Use graph to work out max ΔT during reaction (at 5 minutes)
- Polystyrene is excellent insulator, minimizes heat loss to surroundings
- Allows us to find accurate values for ΔH
- Usually gives accurate results
- Copper calorimeter and spirit burner
- Useful for enthalpy of combustion of fuels
- Heat given out by burning fuel is used to warm a mass of known water
- Measure mass of fuel burned and the ΔT of water
- Allows you to find approximate value for ΔH
- Bomb calorimeter
- Combusts sample in pure oxygen to burn as completely as possible
- Minimises heat loss as much as possible
- Measure mass of sample burned and temperature change of water
Exam Tip You need to know about the different ways of measuring enthalpy change and how to do them |
6. Average bond enthalpy
- The average heat needed when one mole of covalent bonds are broken, measured in the gaseous state
- Average for one type of bond e.g. C-H over lots of compounds
- Bond enthalpies for a type of bond don’t vary much between compounds
- Bond enthalpies for a specific molecule may vary from the average
- Reactions often modelled in three parts
- Bonds of reactants broken
- Atoms rearrange to form products
- Bonds of products form
- In exothermic reactions, bond enthalpies of products is higher than reactants
- Energy is released
- In endothermic reactions, bond enthalpies of products is lower than reactants
- Energy is taken in
- Can use average bond enthalpy data to predict enthalpy change
- ΔH = Σ(bond enthalpies of reactants) - Σ(bond enthalpies of products)
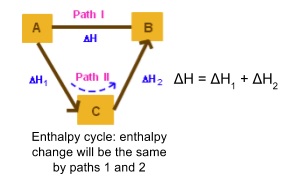
- The enthalpy change of a reaction depends only on the enthalpy of reactants/products
- Doesn’t depend on the route taken
- Can be shown using an enthalpy cycle
- If you know ΔH and ΔH1 for example you can find ΔH2
- Each corner of the cycle has to have the same number of each atom
- Enthalpy changes from enthalpies of combustion
- Given enthalpies of combustion of two substances
- Put them at two corners of the triangle
- Multiply up to have the same number of atoms
- Find the enthalpy change to go from one substance to the other
- Given enthalpies of combustion of two substances
- Enthalpy changes from enthalpies of formation
- Put elements at one corner
- Put compounds of the elements at other corners
- Multiply up to have the same number of atoms
- Add enthalpies of formation and work out enthalpy change from one substance to another
- Put elements at one corner
- For enthalpy changes for unknown reactions you’ll be given the cycle and just need to work out the enthalpy changes
2. reaction rates
1. Concentration and collision theory
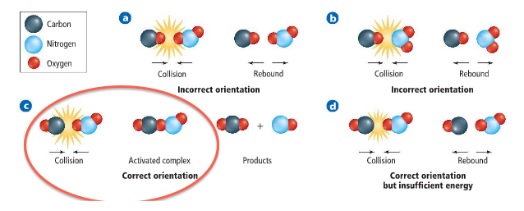
- Collision theory used to explain rates in terms of successful collisions between atoms/molecules
- Atoms/molecules need to collide in the right orientation with enough energy to overcome the activation energy
- As concentration increases there are more particles of reactant per unit volume in solution
- The more particles there are in a solution per unit volume, the greater the chance of collisions between reactants
- So there will be more effective collisions per second with energy in excess of the activation energy
- Same is true for pressure in gases
2. Finding rates from gradients
- Can plot concentration, gas volume of products or mass of substances formed or any other physical quantity that changes during a reaction against time
- Rate at any time is equivalent to the gradient of the graph at that point
- Measure by taking a tangent to the curve
Exam Tip Remember that you find gradient at a point by drawing a flat line against the curved graph, then finding rise over run |
3. Catalysts
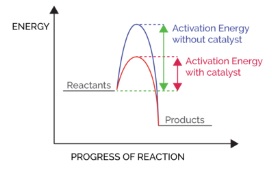
- Catalysts offer an alternative route by which the reaction can happen
- This often means that less energy is required to allow the reaction to take place
- This means that the rate of reaction is faster because more collisions occur with energy in excess of the activation energy
- The catalyst itself is not used up in the reaction, it only speeds up the reaction
4. Types of catalyst and their importance in society
5. Investigating reaction rates experimentally
6. The Boltzmann distribution
- There are homogeneous and heterogeneous catalysts
- Homogeneous
- Same phase as reactants (solid/liquid/gas)
- Enzymes are homogeneous biological catalysts
- Same phase as reactants (solid/liquid/gas)
- Heterogeneous
- Different phase from reactants
- Catalysts in immiscible liquid phases also count as heterogeneous
- Catalytic converters in cars are heterogeneous (solid catalyst, gaseous reactants)
- Different phase from reactants
- Industrial processes strive to have 100% atom economy
- New catalysts developed to improve efficiency
- The industrial production of ethanoic acid has become much more sustainable by the use of a cobalt catalyst
- A process was developed using methanol and carbon monoxide which has an atom economy of 100%
- Scientists then develop a rhodium catalyst, which required lower pressure and temperature
- This has improved yield to around 98% and reduced CO2 emissions
- The industrial production of ethanoic acid has become much more sustainable by the use of a cobalt catalyst
- Catalysts save energy by allowing lower temperatures and pressures to be used reducing CO2 emissions
5. Investigating reaction rates experimentally
- Lots of variables that can be measured over time to follow reaction rate
- Volume of gas produced using a gas syringe
- Measuring mass of a reaction by performing on a balance
- Recording time take for a change to occur e.g. precipitate forming
- Using a colourimeter to measure changes in concentration of a coloured reactant or product
6. The Boltzmann distribution
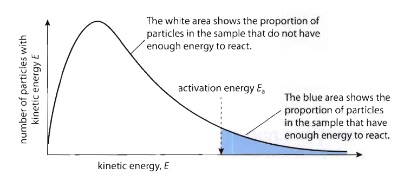
- Distribution of energies (and speeds) of molecules at a particular temperature
- Some molecules are fast with high energy, some are slow with low energy
- Only molecules above the activation energy will react when they collide
- No molecules with zero energy
- No upper limit on energy, just very unlikely for molecules to have really high energy
- Area under the curve represents total number of molecules, doesn’t change with changing conditions
7. Effects of temperature and catalysts on the Boltzmann distribution
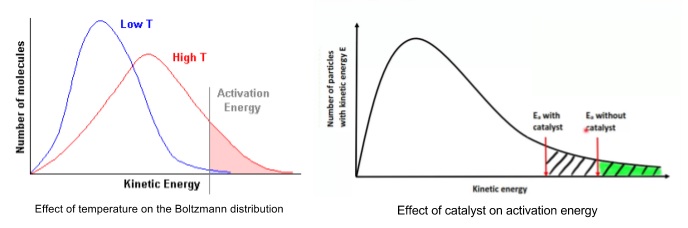
- Temperature shifts the graph to the right and flattens it
- More molecules are above the activation energy so rate increases
- As molecules are moving faster more collisions also occur
- Catalysts provide another pathway with a lower activation energy
- Boltzmann curve shows that there are the same number of particles with the same energy
- But more collisions per second have energy in excess of the new, lower activation energy, so the reaction is faster
3. chemical equilibrium
1. Dynamic equilibrium
1. Dynamic equilibrium
- Some reactions are reversible
- This is represented by the symbol ⇌
- Reactions go both ways
- In dynamic equilibrium the rate of forward (right) reaction is the same as the backward (left) reaction
- Concentrations of products and reactants stay the same
- Dynamic equilibrium can only happen in a closed system
- Position of equilibrium affected by:
- Concentrations of reactants and products
- Pressure of gases
- Temperature
- Effects of changes in conditions predicted by Le Chatelier’s principle
- When a dynamic equilibrium is subjected to change, equilibrium shifts to minimise the change
- Rate of forward or backward increases to minimise change
- Concentration
- Increasing concentration of one species increases the rate of the reaction that consumes that species
- N2(g) + 3H2(g) ⇌ 2NH3(g)
- Increasing H2 concentration increases the forward reaction rate
- Equilibrium shifts to the right
- Pressure
- Only affects equilibrium if gases are present
- When we increase the pressure, the system responds by trying to reduce the pressure
- It does so by moving to the side with fewer particles
- In the reaction above increasing pressure would move to the right
- Increasing pressure of one gas is the same as increasing concentration
- So would move equilibrium away from the side the gas is on
- Temperature
- Increasing temperature causes reaction to take in heat
- Rate of endothermic reaction increases
- N2(g) + 3H2(g) ⇌ 2NH3(g)
- The forward reaction is exothermic
- This means that as result of forward reaction, heat is given out and temperature rises
- If we raise temperature of the reaction then equilibrium will move to the left
- System is opposing change and trying to lower the temperature with the endothermic reaction
- Speed up rates of reaction
- In an equilibrium reaction both forward and reverse rates are speeded up equally
- NO effect on the yield
- We get no more product, we just get it faster
- Lots of different methods
- If coloured compounds are involved colourimeters can be used to measure concentrations
- Titrations can be used to measure concentrations of acids and bases
- Remove samples at set intervals and titrate against known solutions
- Measure pressure of system if gases are involved
- E.g. 2NO2(g) ⇌ N2O4(g)
- NO2 brown, N2O4 colourless
- Mixture of N2O4 and NO2 is put in a syringe
- If compressed, first solution will get darker as concentration increases
- The it will get paler as NO2 is converted into colourless N2O4
- Equilibrium moves to the side with the fewest moles of gas to reduce pressure
- Concentration, this is an example, don’t need to know it specifically
- E.g. ICl + Cl2 ⇌ ICl3
- Iodine chloride can be put in a u-tube
- Chlorine gas is passed over it
- Iodine (III) chlorine, a yellow solid forms
- If more chlorine is added, the concentration is increased
- More yellow solid will form
- Want to get high yield in industry but need to do it in reasonable time
- Haber process good example N2(g) + 3H2(g) ⇌ 2NH3(g)
- Forward reaction is exothermic
- Favoured by high pressure and low temperature
- Low temperature would give low rate so compromise used that balances rate and yield
- High pressure difficult to do industrially
- Requires lots of energy to maintain and can be dangerous so again a compromise pressure is used
- Iron catalyst also used
- Compromise conditions mean only 15% yield achieved but unreacted reactants are easily fed back into the reaction
- Measure of where an equilibrium lies
- For the reaction mA + nB ⇋ pC + qD:
- Kc= [C]p[D]q[A]m[B]n
- Kc > 1 means equilibrium lies to the right, more products formed
- Kc < 1 means equilibrium lies to the left, more reactants formed