AS-LEVEL OCR ChEMISTRY NOTES
TOPIC 2: Electrons, bonding and structure
1. electron structure
- Electron shells
- Electrons in an atom are arranged into shells, or energy levels.
- The shells are numbered with the principal quantum number, n.
- Higher quantum numbers are further from the nucleus and higher in energy
n |
Max number of electrons |
Sub-shells |
1 |
2 |
1s |
2 |
8 |
2s, 2p |
3 |
18 |
3s, 3p, 3d |
4 |
32 |
4s, 4p ,4d, 4f |
2. Orbitals
- Each shell is split into a set of sub-shells, or sub-levels, called orbitals
- An orbital is a region around the nucleus that can hold up to two electrons, with opposite spins
- The first four sets of sub-shell are the s-, p-, d- and f-orbitals
Sub-shell |
Shape of orbital in sub-shell |
Number of orbitals In sub-shell |
Maximum number of electrons in sub-level |
S |
Spherical |
1 |
2 |
P |
Dumbell |
3 |
6 |
D |
- |
5 |
10 |
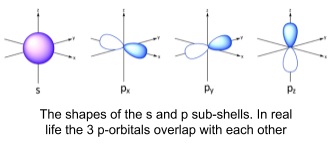
- S orbitals have the lowest possible energy in a shell
- Spherical shape
- As distance from nucleus increases, electron density decreases
- Spherical shape
- P orbitals have the second lowest energy
- They have a node (region of zero electron density) at the nucleus of each atom
- ‘Dumbbell’ shape
- 2 opposite lobes
- They have a node (region of zero electron density) at the nucleus of each atom
Exam Tip The fourth set of sub-shells are called the f-orbitals. You don’t need to know about them, but there are 7 f-orbitals, so it can hold a total of 14 electrons. |
3. Filling orbitals
- Electrons fill up orbitals from the lowest energy first
- The order of energies from lowest to highest is:
- 1s → 2s → 2p → 3s → 3p → 4s → 3d → 4p
- The 4s orbital is lower in energy than the 3d, so it gets filled first
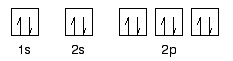
- Orbitals can be represented with box notation
- Each orbital holds two electrons with opposite spins
- Spin is represented by an up or a down arrow
- The diagram represents an atom with 10 electrons
- Electrons prefer to occupy orbitals on their own
- Only pair up when no empty orbitals of the same energy are available
- There are only four electrons in the 2p orbitals below
- Singly fill all three
- Add the remaining electron with an opposite spin
- Singly fill all three
Exam Tip You don’t need to be able to explain spin, just know that electrons in the same orbital have different spins. They’re called spin up and spin down. |
4. Writing electron configurations
- We can write out the electronic configuration (which electrons are in which orbitals) for an atom using sub-shell notation
- Write out the orbitals that have electrons in them in increasing energy order
- Add the number of electrons in each set of orbitals as a superscript next to it
- Sulphur has 16 electrons: 1s2, 2s2, 2p6, 3s2 ,3p4
- Know up to krypton, 36 electrons: 1s2, 2s2, 2p6, 3s2, 3p6, 4s2, 3d10,4p6
- For ions, the highest energy electrons are lost first, so add or take electrons away from the right of the electron configuration for the atom
- Mg: 1s2, 2s2, 2p6, 3s2
- Mg2+: 1s2, 2s2, 2p6
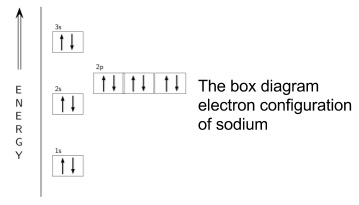
- The electron configuration can also be represented as a box diagram
- These are often done with energy increasing upwards
2. bonding & structure
- Ionic bonding
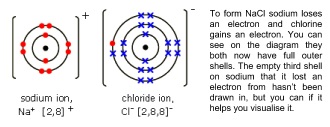
- The most stable electron configurations have a full outer shell
- This is also known as a noble gas configuration, because it’s the same electron configuration that an atom of a noble gas element will have
- Atoms form ions to achieve this, and the ions can bond to each other
- Ionic materials are made up solely of ions, ionically bonded to each other
- An ionic bond is the electrostatic attraction between oppositely charged ions
- The strength of ionic bonding is determined by
- Charges on the ion
- Radii of ions
- Smaller ions form stronger ionic bonds
- Charges on the ion
- Use dot and cross diagrams to show gain and loss of electrons to form ions
Exam Tip Ionic materials are normally made up of a metal that can give up electrons to get a full outer shell and a non-metal that can accept electrons to get a full outer shell |
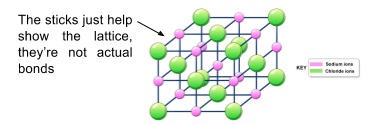
2. Giant ionic lattices
- All ionic compounds have extended structures called giant ionic lattices
- The lattice structure extends in all directions
- Forces exerted by the ions on each other act equally in all directions
- This keeps each individual ion in its place
3. Physical properties of ionic compounds
- The giant lattice structure of ions and the bonding between ions explains the physical properties of ionic compounds
- High melting and boiling points are explained by ions that are strongly held together by the attraction between positive and negative charges
- The electrical conductivity of ionic compounds is explained by whether the ions can move or not
- In solid state the ions are fixed, so ionic compounds don’t conduct
- In liquid and aqueous states the ions can move, so the compounds conduct
- In solid state the ions are fixed, so ionic compounds don’t conduct
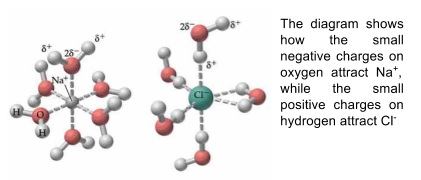
- Ionic compounds dissolve in polar solvents
- A polar solvent is one where each solvent molecule has small negative and positive charges on it
- Ionic compounds are soluble in polar solvents because the charges on the solvent can attract ions from the lattice with opposite charge
- This pulls ions out of the lattice into solution
4. Covalent bonds
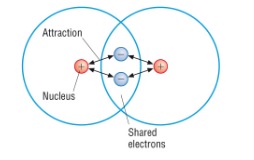
- The strong electrostatic attraction between a shared pair of electrons and the nuclei of the atoms involved in the bond
- Another way atoms achieve full outer shells
- All bonds except for those between atoms of the same element are a mix of ionic and covalent
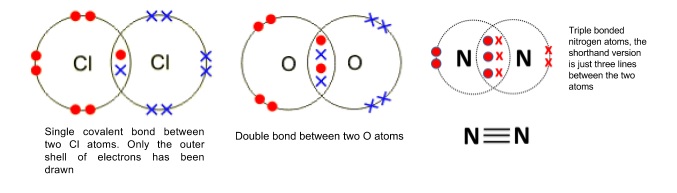
5. Dot and cross diagrams
- Covalent bonds between two atoms are normally represented using dot and cross diagrams
- Normally just draw the outer shell of electrons by drawing one circle around each element
- Use dots to represent electrons from one atom, and crosses for the other
- Overlap the circles of the two bonding atoms and draw the shared electrons in the overlapped part
- Covalent bonds are often drawn as lines between atoms, with double bonds as double lines and triple bonds as triple lines
- Pairs of electrons that aren’t involved in bonding are called lone pairs
- Just drawn on the outer shell of electrons
- Atoms can bond to more than one other atom
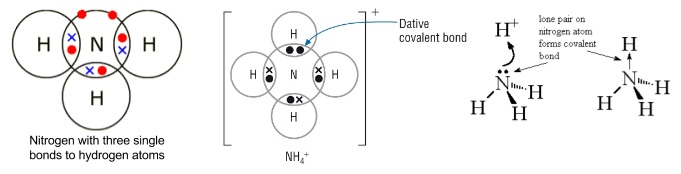
- Atoms can also form dative bonds
- One atom donates both the electrons in a bond
- Drawn as an arrow going from the atom donating electrons to the one receiving them
- Exactly the same as a normal covalent bond once it has formed
- One atom donates both the electrons in a bond
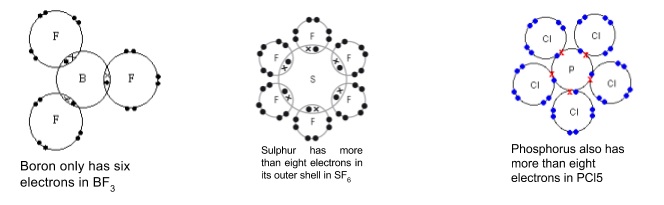
- Some elements don’t have enough electrons to form covalent bonds to get a full outer shell e.g. boron in BF3
- Elements in groups 15-17 from period 3 down have outer shells that can hold more than eight electrons
6. Average bond enthalpy
7. Shapes of molecules and ions
- Measures how much energy is required to break a covalent bond
- The higher the bond enthalpy the stronger the bond
7. Shapes of molecules and ions
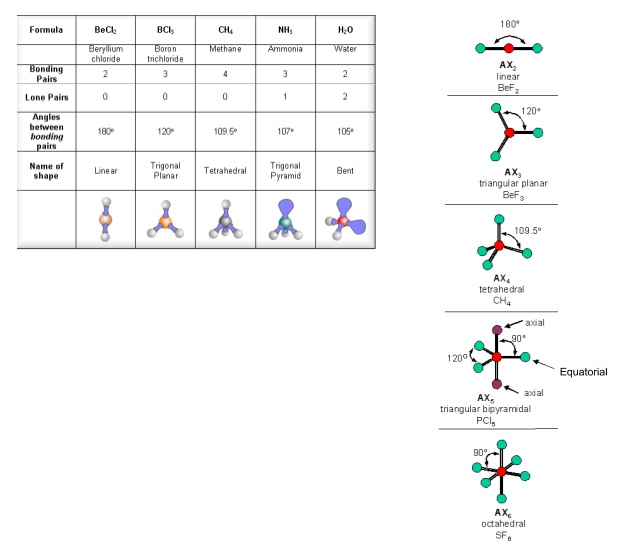
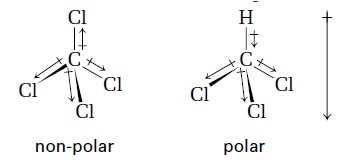
- The shape of a molecule or ion can be predicted by electron pair repulsion theory
- Electron pairs have the same charge so repel other electron pairs
- Arrange electron pairs so they’re as far away from each other as possible
- Lone pairs are more electron dense than bonding pairs so repel more
- So the angle between bonding pairs in CH4 is 109.5o, but in NH3 is 107o, and is only 104.5o in H2O
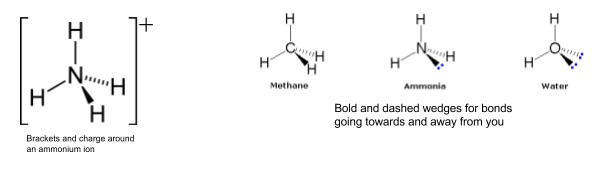
- The shapes of ions can be found in exactly the same way
- Put brackets round the whole ion and add the charge in the top right
- Put brackets round the whole ion and add the charge in the top right
- When drawing 3D shapes use bold and dotted wedges to show bonds going towards and away from you
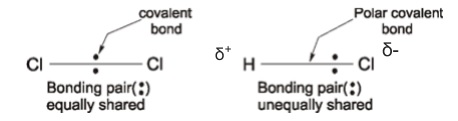
8. Electronegativity
- Covalent bonds can have a degree of ionic character
- Electronegativity is the ability of an atom to attract the bonding electrons in a covalent bond
- Increases going up and to the right on the periodic table
- Measured using the Pauling scale
- The higher an element’s electronegativity, the more strongly it attracts electrons
- Bonds between atoms with the same electronegativity are non-polar
- Bonds between atoms with different electronegativities are polar
- Polar bonds have more of the electron density closer to the more electronegative atom
- The charge difference creates a permanent dipole
- This is shown using δ+ (small positive charge) and δ- (small negative charge)
- E.g. H has an electronegativity of 2.20, and Cl 3.16
- If a molecule has polar bonds it may be polar
- This is when the permanent dipoles of the polar bonds add up to give an overall dipole e.g. CHCl3
9. Intermolecular forces
- Two kinds: weak van der Waals’ forces and stronger hydrogen bonding
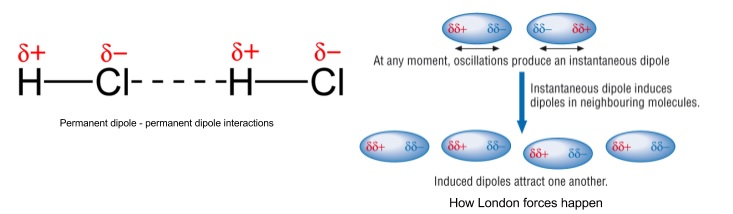
- Van der Waals’ forces include quite weak permanent dipole-induced dipole and permanent dipole-permanent-dipole and weaker London (dispersion) forces
- Permanent dipole-induced dipole is where a molecule with a permanent dipole causes a dipole in a non-polar molecule, the two molecules then attract
- Permanent dipole-permanent dipole is where opposite charges on two molecules with permanent dipoles attract
- London (dispersion) forces is where electrons in atoms are constantly moving
- They become unevenly spread which gives atoms a temporary dipole
- This induces a temporary dipole moment in the neighboring atom, repelling or attracting its cloud
- Attraction is relatively weak, increases with increasing number of electrons
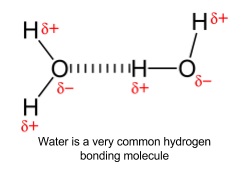
10. Hydrogen bonding
- Occur when H is bonded to N, O or F
- The bond is the attraction between a lone pair on one N, O or F to a δ+ H attached to N, O or F on a nearby molecule
- F, N and O are all very electronegative
- Draw covalent bonding pair of electrons away from the hydrogen
- Bond is polarized
- δ+ hydrogen forms weak bonds with the lone pairs on the other F, N and O
- Stronger than van der Waals’ forces but much weaker than covalent/ionic bonds
- Bond drawn as dashed line
Exam Tip Hydrogen bonding occurs in any molecule with N, O or F bonded to H, so remember it for organic molecules like alcohols e.g. CH3CH2OH (ethanol) |
11. Properties of water
- Hydrogen bonding gives water unusual properties
- Low density
- In ice, molecules are fixed
- Form a fixed lattice of hydrogen bonded molecules
- Hydrogen bonds hold molecules apart
- In liquid water molecules move past each other often
- Form hydrogen bonds less often so less held apart and move closer together
- So ice is less dense than water
- In ice, molecules are fixed
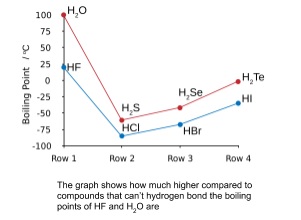
- High melting and boiling point
- Hydrogen bonds are stronger than van der Waals’ forces
- Extra strength has to be overcome to melt or boil water
- So the melting and boiling points are much higher than other compounds like it
- Hydrogen bonds are stronger than van der Waals’ forces
- Also causes things like high surface tension that allows insects to walk on water
12. Simple covalent lattices
13. Physical properties of simple covalent lattices
- Made up of molecules where the atoms in one molecule are bonded together by strong covalent bonds
- Different molecules are bonded by weak intermolecular forces e.g. van der Waals’ forces or hydrogen bonding
- Solid structures have molecules fixed in place by intermolecular forces
- When these change state the forces break
13. Physical properties of simple covalent lattices
- Low melting and boiling points due to weak intermolecular forces
- Non-conductors as there are no free ions
- Mostly soluble in non-polar solvents
- Molecules that form simple covalent lattices are often non-polar
- Like dissolves like
- So they’re soluble in non-polar solvents
- Things like ice that are polar will dissolve in polar solvents