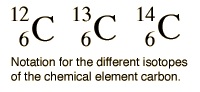AS-LEVEL OCR ChEMISTRY NOTES
TOPIC 1: ATOMS AND REACTIONS
1. ATOMIC STRUCTURE & ISOTOPES
1. Atomic structure
- Atoms are made up of protons, electrons and neutrons
- Neutral atoms have the same number of protons and electrons
- You can work out numbers of protons, electrons and neutrons from the element symbol
- To find the total nuEmber of electrons for positive ions take away the same number of electrons from the atom (e.g. 3 from a 3+ ion) and for negative ions add the same number of electrons
Particle |
Position |
Relative Mass |
Charge |
Proton |
Nucleus |
1.0 |
+1 |
Neutron |
Nucleus |
1.0 |
0 |
Electron |
Shells around nucleus |
1/2000 |
-1 |
2. Isotopes
- Isotopes are atoms of an element with the same number of protons, but different numbers of neutrons
- They have the same numbers of electrons and identical chemical reactions as a result
- The atomic number is often omitted because it stays the same and the element symbol indicates how many protons there are
3. Relative Masses
- The relative isotopic mass is the mass of an atom of an isotope compared with the mass of 1/12th an atom of 12C
- The relative atomic mass, Ar, is the weighted mean mass across all isotopes of an atom compared with 1/12th of the mass of an atom of 12C
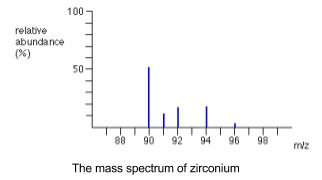
- Mass spectrometry allows the masses of individual molecules or isotopes to be determined by turning substances into positive ions and measuring their mass/charge (m/z) ratio
- Mass spectra plot m/z against percentage abundance
- Number of isotopes can be identified by number of peaks
- Each peak represents an isotope
- To find the relative atomic mass multiply each isotopic mass by percentage abundance, add the results together and divide by 100
- Relative molecular mass
- Relative molecular mass, Mr, is the mass of all the atoms in a molecule of a simple molecular substance
- It’s found by adding up the relative atomic masses of all atoms in a molecule
- Relative formula mass is the mass of one formula unit of a compound that forms a giant structure e.g. NaCl
- It’s found in exactly the same way as Mr
2. COMPOUNDS, FORMULAE & EQUATIONS
1. Ionic formulae
- The formulae of ionic compounds can be found by balancing the charges of the individual ions that make it up
- You need to know the charges of the specific ions and groups of the periodic table shown below
- Find the charges of the ions in a compound and multiply up the number of moles of positive and negative ions until they balance
+1 |
+2 |
+3 |
-3 |
-2 |
-1 |
Group 1 Silver Gold Hydrogen Ammonium (NH4+) |
Group 2 Zinc Tin Lead Iron (II) Copper (II) |
Group 3 Iron (III) |
Group 5 Phosphate (PO43-) |
Group 6 Sulfate (SO42-) Carbonate (CO32-) |
Group 7 Nitrate (NO3-) Hydroxide (OH-) |
Exam Tip Brackets in a formula, like Ca(OH)2, mean the thing in brackets is one ion, and the subscript shows how many |
2. Balancing equations
- Match the number of moles of each element on each side
- To write the ionic equation for reactions involving ions write out just the ions that react
- Leave out all the spectator ions (the ones that don’t change oxidation number or state)
- For ionic equations the charges also need to balance
- Unbalanced, no state symbols: Cu2+ + OH- → Cu(OH)
Exam Tip It’s really easy to forget to put state symbols on during an exam, but you can lose marks for not including them so add them if you’re ever in doubt |
3. Amount of a Substance
1. Moles
- A mole is a measurement of an amount of substance, with the symbol ‘mol’
- It is the amount of any substance containing as many particles as there are carbon atoms in 12g of carbon-12
- Avogadro’s constant, NA, is the number of particles per mole of any substance and is equal to 6.02 x 1023
- The molar mass is the mass per mole of substance in g/mol
Exam Tip The molar volume and NA are in the data sheet, you don’t need to remember them |
2. Empirical and molecular formulae
3. Finding empirical and molecular formulae
- The empirical formula is the simplest whole number ratio of elements in a compound
- The molecular formula is the formula that’s used and is the actual ratio of elements in a compound
3. Finding empirical and molecular formulae
- You can find empirical and molecular formulae for a compound from experimental data
- For the empirical formula, divide the mass of each element by its Ar to find the number of moles of each element present
- Divide by the smallest number of moles
- Multiply up to get whole numbers for each element
- To find the molecular formula, divide the molecular mass (Mr) by the mass of the empirical formula, and multiply the empirical formula by the result
- A hydrated salt is one which contains water of crystallisation e.g. MgSO4·7H2O. An anhydrous salt is one with no water of crystallisation e.g. MgSO4.
- To work out the amount of water of crystallisation in a salt:
- From the Mr - Find the Mr of the anhydrous salt and subtract it from the hydrated Mr then divide the remainder by the Mr of water
- From experimental results - Find the moles of water lost when a hydrated salt is heated in a crucible and the number of moles of anhydrous salt left at the end
- Convert moles, mass and formula mass using moles = massMr
- All gases take up 24 dm3/mol at RTP, this is the molar volume, so n=V24.0mol if V is in dm3
- Divide by 24000 instead if V is in cm3
Exam Tip Sometimes mol/dm3 is abbreviated to ‘M’, they mean exactly the same thing |
6. The ideal gas equation
- Links pressure, P, volume, V, temperature, T, the gas constant, R, and the number of moles of gas present, n.
- pV = nRT
- Use the molar volume to convert masses of gas to volumes
Exam Tip All the quantities in the ideal gas equation need to be in SI units, (p: Pa, V: m3, T:K) so you’ve got to remember to convert any data you’re given. R is in the data book. |
7. Stoichiometric relationships
- The ratios of moles in chemical equations shows how many moles of a product will give how many moles of a reactant and vice versa
- Work out the number of moles of reactants
- Use the ratio to find number of moles of products
- Also works the other way round
- Percentage yield = actual number of moles of products formedtheoretical number of moles of product 100
- Percentage yield may be low due to the reaction not going to completion, impurities or side reactions for example
- Atom economy = molar mass of desired productmolar mass of all products100
- Atom economy will be low if more than one product is formed
- Processes with high atom economies are more sustainable
- They can be productive without depleting resources, as they reduce the amount of raw materials required and reduce waste.
- Addition reactions have atom economies of 100%
- Substitution and elimination reactions have lower atom economies.
4. Acids
1. Acids and bases
- Acids release H+ ions into solution and bases take up H+ ions.
- Alkalis are soluble bases that release OH- ions into solution.
- Common ones you need to know are in the table.
Acid |
Formula |
Salt |
Alkali |
Formula |
Sulfuric |
H2SO4 |
Sulfate |
Potassium Hydroxide |
KOH |
Hydrochloric |
HCl |
Chloride |
Sodium Hydroxide |
NaOH |
Nitric |
HNO3 |
Nitrate |
Ammonia |
NH3 |
Ethanoic/acetic |
CH3COOH |
Acetate |
2. Strong acids
3. Neutralisation reactions
4. Acid-Base Titrations
5. Titration calculations
- Completely dissociate (give up their H+ ions) in solution, weak acids only partially dissociate
3. Neutralisation reactions
- In a neutralisation reaction H+ + OH-(aq)→ H2O(l)
- Acid + Base → Salt + Water
- A salt is an acid with the hydrogen replaced by a metal
- Common bases are carbonates, metal oxides and alkalis e.g. KOH
- Acid + Carbonate → Salt + Water + CO2
- 2HCl(aq) + CaCO3(s) → CaCl2(aq) + CO2(g) + H2O(l)
- Acid + Metal Oxide → Salt + Water
- 2HCl(aq) + MgO(s) → MgCl2(aq) + H2O(l)
- Acid + Alkali → Salt + Water
- HCl(aq) + NaOH(aq) → HCl(aq) + H2O(l)
4. Acid-Base Titrations
- These allow you to find out exactly how much acid is needed to neutralize a quantity of alkali
- Measure alkali into a flask using a pipette
- Add indicator
- Fill burette with known quantity of acid
- Conduct a rough titration to approximate amount
- Add acid form burette
- Swirl flask
- The alkali will be neutralized when the indicator changes colour
- Add acid form burette
- Conduct accurate titration
- Run acid within 2cm3 of the end point
- Slowly add more and record the amount of acid used to neutralize the alkali
- Run acid within 2cm3 of the end point
- Conduct experiment 2/3 times
- Take an average value
5. Titration calculations
- If you know the balanced equation, you can use the results of the titration to find out how much of the unknown alkali (or acid if you’re adding known alkali) is present
- Work out how much acid was added from the volume and concentration
- Use the equation to find the corresponding number of moles of alkali
- Use the number of moles of alkali and the volume to find concentration, or whatever other quantity you need to determine
5. Redox
- Oxidation number
- The oxidation number is the number of electrons an atom uses to bond to atoms of other elements, and is the charge the atom would have if it were fully ionic
- There are a set of rules for working out oxidation number
- Elements bonded to identical atoms have an oxidation number of 0
- The rest are in the table
Species |
Oxidation Number |
Examples |
Uncombined Element |
0 |
S, N2, K |
Combined Oxygen |
-2 |
H2O, Na2O |
Combined Oxygen in Peroxides |
-1 |
H2O2 |
Combined Hydrogen |
+1 |
H2O, NH3 |
Combined Hydrogen in Hydrides |
-1 |
NaH |
Simple Ion |
Ionic charge |
Mg2+, 2+; Na+, 1+; Br-, 1- |
Combined Fluorine |
-1 |
NaF, MgF2, HF |
2. Oxidation numbers in compounds and ions
3. Writing oxidation numbers in compounds
- There are also rules about oxidation numbers for atoms in compounds and ions
- For a complex ion, the sum for each atom must equal overall charge on the ion
- For compounds (ions with a charge of 0), the sum must be 0
- To find the oxidation number for any atom in a compound
- Write out all the oxidation numbers you know, and multiply up by the number of atoms of each type and add them all together
- The remaining unknown oxidation number, when added to the number you just worked out, should equal the overall charge
3. Writing oxidation numbers in compounds
- Some compounds have the same names, but different compositions and oxidation numbers
- Write the oxidation number of the key species as a roman numeral in brackets to make it clear
- For transition metal compounds write the oxidation number of the metal
- FeCl2 = Iron (II) Chloride
- FeCl3 = Iron (III) Chloride
- FeO = Iron (II) Oxide
- Fe2O3 = Iron (III) Oxide
- An oxyanion is an anion that contains an element and oxygen
- Write the oxidation number of the element in the anion that isn’t oxygen
- NaNO2 = Na+ + NO2- = Sodium Nitrate (III)
- NaNO3 = Na+ + NO3- = Sodium Nitrate (V)
- SO32- = Sulfate (IV)
- SO42- = Sulfate (VI)
Exam Tip If you’re asked to write down the name of one of these compounds from the formula, you need to put the oxidation number in or you could lose marks |
4. Redox reactions
5.Redox reactions with metals and acids
- Redox reactions are where one species is reduced and another is oxidised
- Oxidation is loss of electrons
- Reduction is gain of electrons
- The species that is reduced takes electrons from the one that is oxidised
- Redox can also be understood in terms of oxidation number changes
- If something is oxidised its oxidation number increases by 1 for each electron lost
- If something is reduced its oxidation number decreases by 1 for each electron gained
- Metals tend to get oxidised, while non-metals tend to get reduced
5.Redox reactions with metals and acids
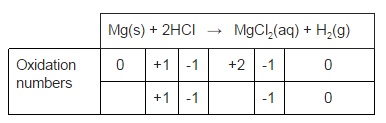
- Metal + Acid → Salt + Hydrogen
- Mg(s) + 2HCl(aq) → MgCl2(aq) + H2(g)
- Assign oxidation numbers to each species in the reaction to work out what’s being reduced or oxidised
- Mg has gone from 0 → +2 so is oxidised
- H has gone from +1 → 0 so is reduced
6. Unfamiliar redox equations
- Put in the oxidation numbers for all the species on both sides of the equations first
- Find the species that change oxidation number
- Assign them as reduction if it’s decreased and oxidation if it’s increased
Exam Tip The total change in oxidation number will always be zero, so you can add up all the changes you’ve found to check if you’ve got everything. |