AS-LEVEL OCR ChEMISTRY NOTES
TOPIC 1: basic concepts and hydrocarbons
1. Basic concepts of organic chemistry
1. IUPAC nomenclature for systematic naming of compounds
- Specific naming system used to give unambiguous names to compounds
- Three parts to a name: stem, suffix and prefix
- Stem: main part, depends on the longest carbon chain in a molecule
- Based on name of the alkane that corresponds to the longest carbon chain
Alkane |
Number of C atoms in the main carbon chain |
Methane |
1 |
Ethane |
2 |
Propane |
3 |
Butane |
4 |
Pentane |
5 |
Hexane |
6 |
Heptane |
7 |
Octane |
8 |
Nonane |
9 |
Decane |
10 |
- Suffix: end of the name, identifies the most important functional group
- If a suffix starts with a vowel the -e at the end of the alkane stem is dropped
- Prefix: start of the name, identifies any other functional groups and the carbons they’re attached to
Functional group |
Formula |
Prefix |
Suffix |
Alkanes |
C-C |
-ane |
|
Alkenes |
C=C |
-ene |
|
Haloalkanes |
-F -Cl -Br -I |
fluoro- chloro- bromo- iodo- |
|
Alcohols |
-OH |
hydroxy- |
-ol |
Carboxylic Acid |
-COOH |
-oic acid |
|
Aldehydes |
-CHO |
-al |
|
Ketones |
C-CO-C |
-one |
- Numbers are used to indicate the location of a carbon side chain or functional group on the main chain
- Dashes separate numbers from words
- Commas separate numbers
- To name a compound
- Identify the longest carbon chain and the right alkane stem
- Identify the most important functional group and which carbon on the carbon chain it is attached to
- Attach the suffix to the stem with number of the carbon it’s on before it, removing the -e from the stem if needed
- Identify any other functional groups and add them as prefixes
- Prefixes are written in alphabetical order, numbering the carbons so that the smallest numbers possible are used
- di- indicates two identical functional groups, tri- three and tetra- four
- Identify the longest carbon chain and the right alkane stem
- Aldehydes and carboxylic acids don’t need numbers because they’re always at the end of a chain
- Alkanes with a hydrogen removed are called alkyl groups
- They’re added as prefixes when attached to the main carbon chain
Alkyl group |
Number of C atoms in the side carbon chain |
Methyl |
1 |
Ethyl |
2 |
Propyl |
3 |
Butyl |
4 |
Pentyl |
5 |
Hexyl |
6 |
Heptyl |
7 |
Octyl |
8 |
Nonyl |
9 |
Decyl |
10 |
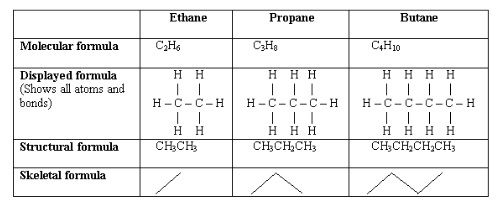
2. Interpreting structures
- General formula: the simplest algebraic formula of a member of a homologous series, represents the actual number of atoms in each molecule e.g. C4H10
- Structural formula: shows number of atoms present and way in which they are arranged relative to each other
- Displayed formula: shows actual places of the atoms and nature of the bonds between them
- Skeletal formula: carbon and hydrogen atoms omitted with carbon atoms represented by junctions between bonds
3. Functional groups and homologous series
4. Formulae for homologous series
- Functional group: a group of atoms responsible for the characteristic reactions of a compound
- Homologous series: a series of organic compounds having the same functional group but with each successive member differing by CH2
- Homologous series generally have similar chemical properties
- Their physical properties change as each unit of CH2 is added
- Their molecular formula can be worked out from a basic formula for the series
- Alkyl group: an alkane missing a single hydrogen, can be a side chain on other carbon chains
- Formula CnH2n+1 e.g. propyl CH3CH2CH2 (C3H7)
- Aliphatic: a compound containing carbon and hydrogen joined together in straight chains, branched chains or non-aromatic rings e.g. 2,3-dimethylpentane
- Alicyclic: an aliphatic compound arranged in non-aromatic rings with or without side chains e.g. cyclohexane
- Aromatic: a compound containing a benzene ring
- Saturated: a compound with single carbon–carbon bonds only
- Unsaturated: the presence of multiple carbon–carbon bonds, including C=C, triple bonds and aromatic rings
4. Formulae for homologous series
- Each homologous series has a general formula that lets you work out the molecular formula for a given number of carbons
- E.g. alkanes CnH2n+2, alkenes CnH2n, alcohols CnH2n+1OH
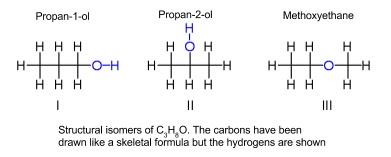
- Compounds can have the same molecular formulas, but different structural formulas
- These are structural isomers
- Three ways for isomers to be different
- Alkyl groups in different places
- Functional groups in different places
- Functional groups rearranged into different functional groups
6. Reaction mechanisms
7. Radicals
- Covalent bond fission (breaking) can happen in two ways
- Heterolytic Fission
- Bond breaks unevenly
- Both electrons from shared pair move to one atom
- Bond breaks unevenly
- This forms two species:
- Positively charged cation (electrohpile)
- Negatively charged anion (nucleophile)
- Positively charged cation (electrohpile)
- Homolytic Fission
- Bond breaks evenly
- One electron moves to each atom
- This forms two electrically uncharged radicals
- Bond breaks evenly
7. Radicals
- Formed in homolytic fission
- Have an unpaired electron
- Because of the unpaired electron, radicals are very reactive
- Represented in reaction mechanisms by a single dot next to the radical
Exam Tip In drawn out reaction mechanisms, radical unpaired electron movements are shown by a half curly arrow with only one ‘hook’, but you don’t need to know how to do these because all the examples you’ll come across will just be equations like the one above |
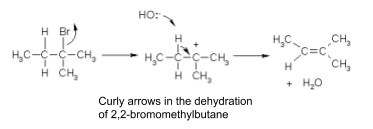
8. Curly arrows
- Used to show the movement of a pair of electrons in a reaction mechanism
- Happens in either heterolytic fission (going to the atom that becomes negatively charged) or covalent bond making
- Start from a bond, a lone pair or a negative charge
- Point to exactly where the electrons are going
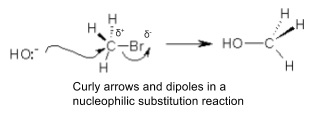
9. Reaction mechanisms
- Models of how electrons move during a reaction
- Curly arrows show movement of electrons in each step
- Show actual charges with small plus or minus signs next to the atom that has the charge like carbon in the mechanism above
- Show dipoles on important bonds where the two atoms have different electronegativities
2. alkanes
1. Alkanes
2. Bonding in alkanes
- Alkanes are saturated hydrocarbons
- This means that they contain only carbon to carbon single bonds
- Carbon has four electrons in the outer shell (group 14) so forms four bonds
- General formula of CnH2n + 2
- Each C-C or C-H bond is formed from direct overlap of orbitals of bonding atoms
- This is called a sigma (σ) bond
- These can freely rotate around the axis of the bond
2. Bonding in alkanes
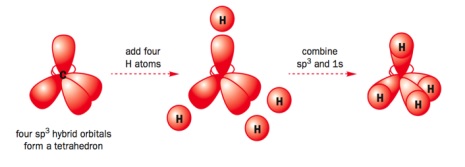
- The carbon 2s and three 2p orbitals combine to form four hybrid sp3 orbitals
- These are what overlap to form sigma bonds
- They are shaped like lopsided p orbitals
- Arranged in a tetrahedral formation 109.5o away from each other
- So carbon atoms in alkanes have the atoms they’re bonded to arranged in a tetrahedral formation
- This is explained by electron pair repulsion theory as it allows the electrons in the bonds to be as far away from each other as possible
3. Boiling/melting points in alkanes
4. The low reactivity of alkanes
- Alkanes have covalent bonds inside molecules
- They have London forces between molecules, holding them together
- The longer the carbon chain, the stronger the forces
- There is more molecular surface area and each molecule has more electrons that can interact
- So short chain alkanes have lower boiling points than long chain alkanes
- Effect of branching
- Branched-chain alkanes have smaller molecular surface areas
- They are not as closely packed
- London forces are reduced
- Branched chain alkanes have lower boiling points than straight chain alkanes
4. The low reactivity of alkanes
- All covalent sigma bonds have high enthalpies so are hard to break
- Carbon and hydrogen have very similar electronegativities so the bonds have very low polarities
- This means they aren’t reactive to electrophiles/nucleophiles either
- Combustion (burning) is a rapid oxidation reaction which combines oxygen with another substance
- All hydrocarbons undergo complete combustion when plenty of O2 is present with a clean blue flame to give CO2 and H2O
- High numbers of carbon and hydrogen atoms mean hydrocarbons burn well and release lots of heat energy
- E.g. burning methane (used in domestic cooking and heating)
- CH4 + 2O2 → CO2 + 2H2O
- Methane is the main gas used in domestic heating and cooking
- If there is insufficient oxygen, then the alkanes undergo incomplete combustion
- This produces colourless, odourless carbon monoxide, CO, and soot, C, with a yellow flame
- Lots of different reactions e.g.
- 2CH4 + 2½O2 → CO + C + 4H2O
- 2CH4 + 3½ O2 → CO2 + CO + 4H2O
- Carbon monoxide is toxic, because it reduces the capacity of blood to carry oxygen
- If appliances are not properly maintained, then insufficient oxygen may be supplied to the burners
- This may cause carbon monoxide to be produced
- Alkanes react with halogens in UV light to form haloalkanes
- Reaction process proceeds by a free radical mechanism with three stages
- Initiation - formation of radicals
- Cl2 → 2Cl⠂
- Needs UV or ~300oc
- Propagation - two repeated steps that build up product in a side reaction
- CH4 + Cl⠂→⠐CH3 + HCl (making alkyl radical)
- ⠐CH3 + Cl2 → CH3Cl + Cl⠂(forming haloalkane)
- Termination - two radicals react to form a stable product
- Lots of possible reactions
- 2Cl⠂→ Cl2
- 2⠐CH3 → C2H6
Exam Tip You need to know radical substitution reactions and their three stages for both chlorine and bromine, but they both happen in exactly the same way |
7. Limitations of radical substitutions
- Because so many different radicals can be formed in the propagation and termination steps, there are lots of different possible products
- Further products can be formed if the products of one chain are involved in more propagation steps
- Therefore there’s a low atom economy and yield with lots of undesired side products
3. alkenes
1. Alkenes
- Alkenes are a homologous series of unsaturated hydrocarbons
- General formula of CnH2n
- Unsaturated means that they contain double carbon to carbon bonds
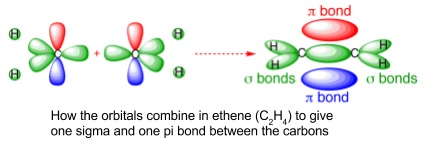
- Made up of one sigma and one pi (π) bond
- π-bond is formed by sideways overlap of p-orbitals compared to head on overlap for σ-bonds
- π-bonds have 2 regions of electron density, above and below the internuclear axis
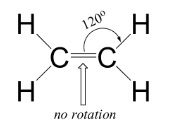
- They prevent rotation about the bond because the two p-orbitals have to be parallel to each other
- This means the alkene is flat in the region of the double bond
2. Trigonal planar carbon atoms in alkenes
- Each carbon has three other atoms bonded to it around it
- Two hydrogens σ-bonded to it
- One carbon σ- and π-bonded to it
- The σ-bonds stay 120o apart to keep the electrons in the bonds as far away from each other as possible (electron pair repulsion theory)
- So the carbons have a trigonal planar geometry around them
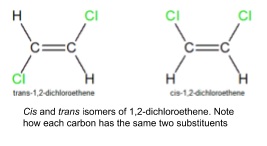
3. Stereoisomerism in alkenes
- Stereoisomers are compounds with the same structural formula but with a different arrangement of the atoms in space
- There can be stereoisomers of alkenes because the double bond can’t rotate
- Rotation would require the breaking of the pi bond, which requires a great deal of energy
- For an alkene to show geometric isomerism, there must be two different types of atoms attached to each carbon in the double bond
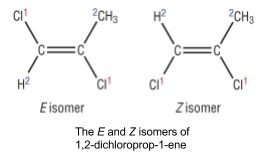
- Alkenes have E and Z isomers
- Look at one carbon atom of a double bond and assign priority using Cahn-Ingold-Prelog rules to the substituents
- The highest molecular weight substituent has the highest priority and so on
- Do the same for the other carbon atom
- If the highest priority substituents for each carbon are on the same side of the double bond it’s the Z-isomer
- Cis-trans naming is appropriate if each carbon has two identical groups attached to it
- Split the alkene along the line of the carbon-carbon double bond
- If both sides have the same group it’s the cis-isomer
- Split the alkene along the line of the carbon-carbon double bond
4. Stereoisomers from structural formulas
5. Reactivity and the π-bond
6. Electrophiles
7. Addition reactions of alkenes
- Given a structural formula with a double bond, work out which isomer it is
- Then swap constituents at the double bond to get the other isomer
5. Reactivity and the π-bond
- The π-bond has a lower bond enthalpy than the σ-bond
- This means it’s easier to break
- So alkenes are more reactive than alkanes
6. Electrophiles
- Species that are electron-pair acceptors
- Could be because it’s a positive ion or has a δ+ region
- Attracted to the high electron density in π-bonds
7. Addition reactions of alkenes
- A reaction where the π-bond breaks and a small molecule is added across the two carbon atoms
- Gives a saturated organic product
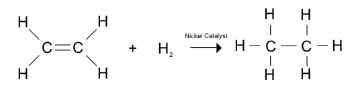
- Hydrogenation is the addition of an H2 molecule across the double bond
- Suitable catalyst e.g. nickel
- Suitable catalyst e.g. nickel
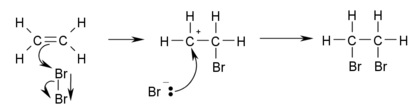
- Halogenation is the addition of a halogen (Cl2, Br2 etc) across the double bond
- Reactions proceed at room temperature
- No UV light required
- Forms di-substituted haloalkanes
- In the first step a dipole is induced in Br2 as it approaches the electron rich double bond
- Makes the closer Br δ+
- This then attracts electrons from the double bond
- Reactions proceed at room temperature
- Test for saturation
- If an organic compound decolourises bromine or iodine solution a double bond is present
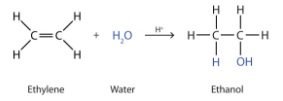
- Hydration is the addition reaction of water across the double bond of a gaseous alkene to make an alcohol
- Temperature of 300oc
- Pressure of 65 atm
- Temperature of 300oc
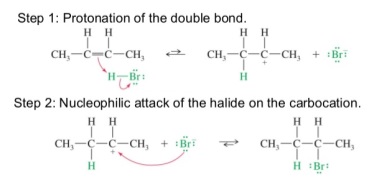
- Hydrogen halides, HX, (HCl, HBr etc) add across the double bond to give haloalkanes
- Hydrogen halides are gases
- Bubbled through alkenes to give a reaction
- Hydrogen halides are gases
Exam Tip You need to know the mechanisms for all these addition reactions |
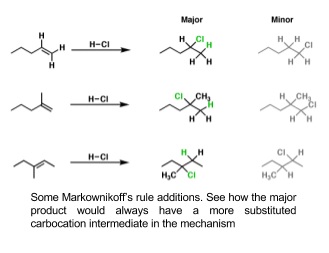
8. HX adding to unsymmetrical haloalkanes
- If the alkene is unsymmetrical two products can form
- Use Markownikoff’s rule to work out the major product
- The major product is the molecule in which hydrogen adds to the carbon with the greatest number of hydrogens already attached to it
- This is related to the stability of the carbocation intermediate formed
- Stability: tertiary carbocation > secondary carbocation > primary carbocation
- More alkyl groups attached to a carbon make the carbocation more stable
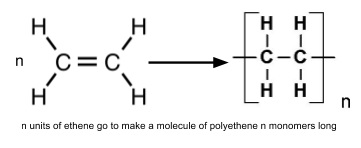
9. Addition polymerisation of alkenes
- Polymers are large molecules made from smaller monomer units
- The double bond in alkenes ‘opens up’ to form polymers
- π-bond breaks and the carbon atoms form σ-bonds to other monomers
- Polymers can be millions of monomers long so we draw a repeating unit
- To make a repeat unit from a monomer
- Draw the individual monomer in square brackets
- Turn the double bond into a single bond
- Add bonds on either side extending out of the square brackets
- Add subscript n, this shows how many monomers are in a polymer molecule (can be very large)
- Draw the individual monomer in square brackets
- To find a monomer from a polymer
- Choose a group of atoms and find where it repeats
- The section between the two repeat points is the repeat unit
- Make sure it’s as small as possible i.e. one monomer not two
- Do the above steps in reverse to get the monomer molecule
- Choose a group of atoms and find where it repeats
Exam Tip You need to be able to find monomers from polymers and create polymers from monomers |
10. Sustainability and processing waste polymers
11. Biodegradable polymers
- Plastics and synthetic polymers are non-biodegradable
- Cannot be broken down by living organisms in the way that natural compounds are
- Waste plastics that are sent to landfill and remain there for many years as they only decompose slowly
- Plastics are mainly hydrocarbon so can be burned
- This allows some of the energy used to make them to be recovered
- But this also produces CO2
- Some plastics produce toxic waste products e.g. HCl when burned
- These can be removed using gas scrubbers
- This allows some of the energy used to make them to be recovered
- Plastics can be recycled by being melted down and reshaped into new things
- Plastics can also be broken down to small organic molecules to be used in other polymers or industrial reaction
- This reduces the need for plastic disposal and the demand for the production of plastics
- Recycling polymers saves the energy that would be used making new polymers
- Only uses the energy needed for the process of recycling
- But:
- The plastics need to be sorted to be recycled
- This takes time or expensive equipment
- There’s a small market for recycled plastic because companies are concerned about quality and contamination from previous use
- The plastics need to be sorted to be recycled
11. Biodegradable polymers
- Polymers that can be attacked by microorganisms and environmental conditions and broken down into harmless or useful substance
- Should break down in the same amount of time as normal green waste
- Biodegradable polymers can be mixed with addition polymers like polyethene
- The biodegradable polymer can break down allowing the material to biodegrade
- This leaves small pieces of plastic which can also be dangerous
- If just biodegradable polymers are used it’s a bioplastic
- These fully degrade so don’t create landfill or need energy to be recycled
- They can be carbon neutral if they’re made with sustainable farming methods
- Photodegradable polymers break down in sunlight
- This reduces the chance they damage the environment
- Once exposed to light it’s not possible to stop this process
- They might not get exposed to enough light in a landfill for this to happen