AS-LEVEL OCR ChEMISTRY NOTES
TOPIC 2: alcoholsm, haloalkanes and analysis
1. alcohols
1. Properties of alcohols
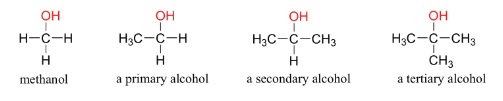
- Saturated, homologous series with formula CnH2n+1OH
- Can be primary (1 C attached to OH C), secondary (2 Cs attached to OH C) or tertiary (3 Cs attached to OH C)
- Are polar because the oxygen atom is more electronegative than carbon
- Have relatively low volatility
- Volatility is a measure of how easily molecules can escape from a liquid
- A volatile liquid loses molecules from the surface very easily and so it will have a low boiling temperature
- Branching generally decreases boiling point, and so increases volatility
- Alkanes have a much lower boiling point than an alcohol with the same number of carbons
- In alkanes the intermolecular forces are only weak induced dipoles (London Forces)
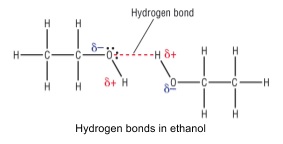
- In alcohols, the OH group can form hydrogen bonds, which are much stronger
- These hydrogen bonds require much more energy to be broken
- So boiling points are higher
- Alcohols are covalent but dissolve in water
- Polar OH group in alcohol forms hydrogen bonds with polar OH group in water
- Carbon chain can’t form hydrogen bonds to water
- The longer the carbon chain, the less soluble the alcohol is
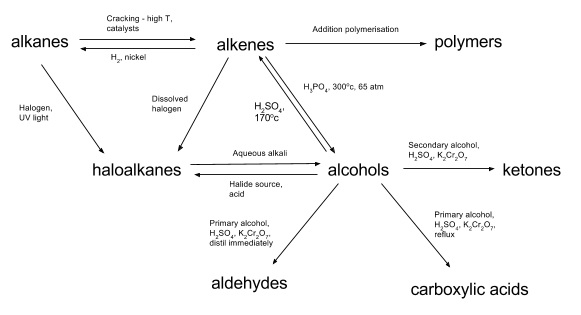
2. Combustion of alcohols
- Happens in the same way as for alkanes producing CO2 and water
- e.g. C2H5OH(l) + 3O2(g) → 2CO2(g) + 3H2O(l)
- Oxidation of the –OH group to C=O (carbonyl) group
- Acidified potassium dichromate (VI) usually used as oxidising agent (H+/K2Cr2O7)
- Represented by [O] in equations
- Cr (VI) is reduced to Cr3+ ions
- Colour change from orange to green
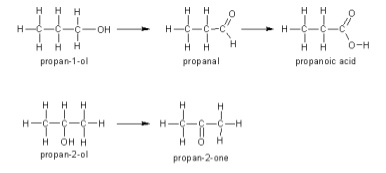
- Functional group of product depends on whether it is a primary, secondary or tertiary alcohol
- Primary
- Oxidised to form aldehydes
- Reaction stops if aldehyde is distilled off straight away
- Reaction will continue if heated under reflux
- These are then oxidized to form carboxylic acids
- Secondary
- Heat under reflux
- Oxidised to form ketones
- Tertiary
- These cannot be oxidised
- No reaction occurs
4. Dehydration of alcohols
- H2O can be eliminated from alcohols to give alkenes
- Heat under reflux with a strong acid e.g. H2SO4/H3PO4
- The acid is a catalyst for the reaction
Exam Tip An elimination is a reaction where one molecule forms two products, and normally one is small. A dehydration is an elimination where water is one of the products |
5. Substitution with halide ions
- Alcohols undergo a nucleophillic substitution reaction with halide ions
- -OH group replaced by Cl-/Br-/I-
- ROH + HX → RX + H2O
- An acid catalyst is added along with the halide source and the mixture is warmed
- To form a chloroalkane, HCl is added
- To form a bromoalkane, normally NaBr is added with H2SO4 to make HBr in situ
- This doesn’t work for iodine as it’s oxidized by H2SO4 to give I2 so the yield is poor
- Phosphoric acid is used instead
Exam Tip You don’t need to know a mechanism for this |
2. haloalkanes
1. Nucleophiles
2. Hydrolysis of haloalkanes
- Electron pair donors (and also Lewis bases)
- Have partial (δ-) or full negative charges
- Attracted to positive charges or δ+
- E.g. H2O, NH3, OH-
2. Hydrolysis of haloalkanes
- Haloalkanes are alkanes with one or more hydrogen replaced by a halogen
- There’s a large difference in electronegativity between carbon and the halogens
- So the bond is polar, carbon is δ+ and the halogen δ-
- Haloalkanes undergo hydrolysis
- This is a substitution reaction with water as a nucleophile (nucleophilic substitution)
- Hydrolysis with aqueous alkali to form alcohols
- CX + OH- → COH + X-
- -OH group substituted for the halogen
- NaOH or KOH used
- Done under reflux conditions
- -OH group substituted for the halogen
- Haloalkanes can also react with AgNO3 and water
- Halide ion leaves during the reaction
- This reacts with Ag+ to give a precipitate
- Can use how long the precipitate takes to appear to determine how fast reactions of different haloalkanes are
- Add 1cm3 of each haloalkane to different test tubes in a water bath at 50oc
- Put test tubes with ethanol, water and aqueous silver nitrate in the water bath
- Allow them to come to the same temperature
- Add 1cm3 of ethanol, water and aqueous silver nitrate to each haloalkane
- See how long it takes for precipitate to appear
- Add 1cm3 of each haloalkane to different test tubes in a water bath at 50oc
- Use this to determine bond strengths of different C-X bonds
- Stronger bonds will lead to a slower reaction so precipitate will take longer to appear
Haloalkane |
Halide |
Colour of AgX precipitate |
chloroalkane |
Cl- |
White |
bromoalkane |
Br- |
Cream |
iodoalkane |
I- |
Yellow |
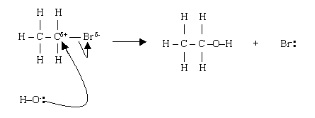
3. The mechanism of nucleophilic substitution
- Electrophilic OH- attacks δ+ carbon
- Reaction shows bromine but could be any halogen
4. Hydrolysis rates of primary haloalkanes
5. Haloalkanes and the environment
- Explained by C-X bond enthalpies
- Going down halogens atoms increase in size
- Bonding electrons are further from nucleus so more shielded
- This gives lower C-X bond enthalpies
- Easier to break the bond so reaction happen faster
- Electronegativity increases up the group
- C-F bonds are most polar so attract nucleophiles more
- But bond enthalpies are very high so hardest to break and react slowest
5. Haloalkanes and the environment
- Haloalkanes used in industry as CFCs
- CFCs are gases and eventually escape into the atmosphere
- Exposed to UV light in the stratosphere
- C-X bonds break in UV light
- Undergo homolytic fission to form radicals
- E.g. CHFCl2 → CHFCl⠂+ Cl⠂
- Ozone (O3) is present in the stratosphere as the ozone layer (20km up)
- Protects earth from UV which causes cell mutations responsible for cancer and cataracts by absorbing it
- Radicals including from CFCs catalytically destroy ozone e.g.
- Propagation: Cl⠂+ O3 → ⠐ClO +O2
- Propagation:⠐ClO + O3 → Cl⠂+ O2
- Overall: 2O3 → 3O2
- Propagation: Cl⠂+ O3 → ⠐ClO +O2
- Also happens with other radicals e.g. ⠐NO
- Propagation:⠐NO + O3 → ⠐NO2 +O2
- Propagation:⠐NO2 + O →⠐NO + O2
- Propagation:⠐NO + O3 → ⠐NO2 +O2
3. organic synthesis
1. Practical skills
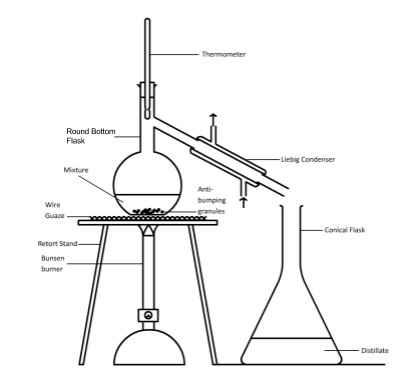
- Quickfit apparatus used in practical organic chemistry
- Grease joints with petroleum jelly to get a good fit or use plastic connectors
- Distillation is a separating technique used to collect product from an organic synthesis reaction
- Equipment
- Round bottom flask for reaction mixture
- Heating mantle
- Thermometer and holder
- Condenser and connector to join it to the round bottom flask
- Collecting flask
- Round bottom flask for reaction mixture
- Set up using clamp stands and bosses to hold equipment
- Connect bottom of condenser to tap with rubber hose and run rubber hose from top of condenser to sink and turn on gentle flow of water
- Pour reaction mixture into flask with anti bumping granules
- Heat and note temperature that products starts evaporating and being collected
- This is its boiling point and helps to identify it
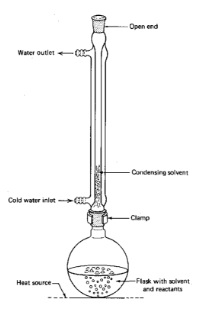
- Heating under reflux is a technique used to ensure that volatile compounds in a reaction mixture don’t boil away
- Equipment
- Pear shaped flask
- Heating mantle
- Condenser
- Pear shaped flask
2. Preparing and purifying organic liquids
3. Identifying functional groups and predicting properties
- Many organic liquids are immiscible in water
- Can use a separating funnel to separate organic product from an aqueous solvent
- Mount an iron ring on a clamp stand and put separating funnel in it
- Ensure tap is closed and add reaction mixture until no more than half full
- Wash reaction vessel with water, add to funnel and put stopper on
- Take funnel out of ring and invert, open tap to equalise pressure, close tap
- Gently shake mixture, opening tap every now and then while inverted to release pressure
- Carry on until no longer hear ‘whistle’ when opening the tap
- Put funnel in iron ring and allow layers to seperate
- Remove stopper, put a beaker under the spout and open tap
- Collect the lower water layer in the beaker and turn off the tap
- Aqueous layer can be discarded as it has no product in it
- Place fresh beaker under tap and collect product
- Shake with drying agent and pour product into fresh container
- Drying agents are anhydrous inorganic salts like MgSO4 and CaCl2
- They absorb water so can dry other liquids
- Add a few spatulas of drying agent to wet organic product
- If the drying agent clumps add more
- When the drying agent remains free moving the organic product is dry
- Use gravity filtration to remove drying agent and leave dry product
- Add a few spatulas of drying agent to wet organic product
- Redistillation purifies product through multiple distillations
- Keep repeating a distillation, keeping only the product each time, until desired purity is achieved
3. Identifying functional groups and predicting properties
- Need to be able to recognise double bonds, haloalkanes, carbonyls (C=O), carboxylic acids and alcohols
Functional group |
Reagents to test for |
Observations |
unsaturated hydrocarbon (C=C) |
Add a few drops of bromine water and shake |
Bromine water decolourises |
haloalkane |
Silver nitrate, ethanol, water |
White precipitate = chloro- Cream precipitate = bromo- Yellow precipitate = iodo- |
carbonyl |
Acidified potassium dichromate (VI) Fehling’s solution Tollens’ reagent |
Ketones - no change Aldehydes - orange → green Ketones - no change Aldehydes - dark red precipitate Ketones - no silver mirror Aldehydes - silver mirror |
carboxylic acid |
Universal indicator or pH probe Reactive metal e.g. Ca Carbonate e.g. MgCO3 |
pH of a weak acid Hydrogen effervesced CO2 effervesced |
alcohol |
Warm with equal volume of carboxylic acid and a few drops of H2SO4 |
Sweet smell of an ester after a short time |
- Many compounds have more than one functional group
- These can all be identified individually
- Properties will all be dependent on what functional groups are present, and can be predicted from the properties of simple examples of the functional groups shown
4. Synthetic routes
- Need to be able to give two step reactions that generate one compound from another
4. analytical techniques
1. Infrared radiation
2. Absorption of IR radiation by atmospheric gases
3. Identifying functional groups in IR spectra
- Different bonds in covalent molecules absorb radiation at particular frequencies
- Frequency of infrared radiation depends on the energy required to cause the bond to stretch or bend
- Amount of vibration depends on:
- Bond strength
- Bond length
- Mass of each atom involved in the bond
- Bond strength
- Infrared spectra usually have a range from 300 to 4000cm-1
2. Absorption of IR radiation by atmospheric gases
- IR absorption happens everywhere, not just inside spectrometers
- C=O, O-H and C-H bonds are very good at absorbing IR
- These bonds are found in common greenhouse gases like CO2, H2O, CH4
- These gases absorb IR radiation leaving the earth and emit it back towards the earth
- This is the greenhouse effect
- These gases are suspected of causing global warming
- So governments have introduced policies to limit their emissions and encourage technologies and energy resources that don’t emit them
3. Identifying functional groups in IR spectra
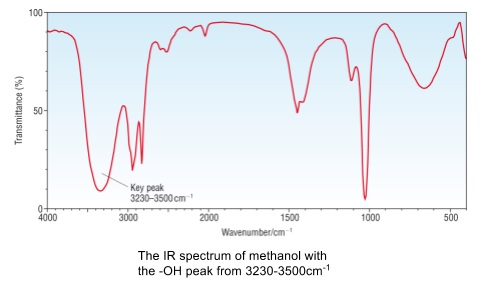
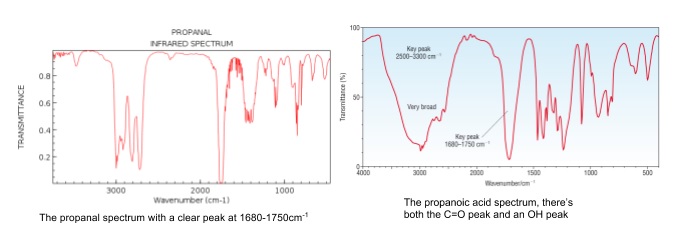
- O-H bond in alcohols has a broad absorption at 3200-3550cm-1
- If this peak is present the compound being analysed has an alcohol group
- Most organic compounds have an absorption at ~3000cm-1 due to C-H bonds
- Don’t confuse this with a broad O-H peak
- In the spectra below these are the peaks just to the right of the O-H peak
- C=O bond in aldehydes, ketones and carboxylic acids absorbs from 1640-1750cm-1
- If no OH peak is present, the compound is an aldehyde/ketone
- If an OH peak is present, the compound is a carboxylic acid
Exam Tip Infrared absorption data is given in the data sheet, this will tell you what bonds absorb at what frequencies |
4. Interpretation of spectra
5. Uses of IR spectroscopy
6. Mass spectrometry
- If you’re given a spectra, identify what clear peaks are present
- Match these up with the known absorptions of functional groups
- Use these to work out what functional groups are in the molecule
5. Uses of IR spectroscopy
- Used in monitoring air pollution
- Pollutants like CO and NO have specific absorptions that allow their concentrations to be monitored
- This can be used to study the effect of traffic on air quality
- Also used in breathalyzers
- The strength of the absorption peak of alcohols in breath depends on the concentration in the blood
- This phenomenon is described by the Beer-Lambert law
- So IR spectra can be used to determine blood alcohol levels
- Commonly used alongside other technologies like fuel cells that react with alcohols or chemical reactions that show the presence of alcohols to identify alcohol
6. Mass spectrometry
- Mass spectrometers only detect ions
- Organic compounds vapourized and ionized when driven through a mass spectrometer
- Some form a molecular ion, M+
- This is when the molecule loses an electron
- The mass of the electron is negligible, so this has the same mass as the relative molecular mass of the original molecule
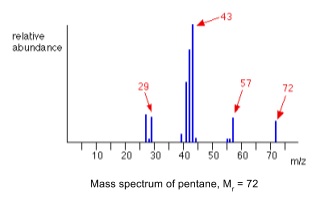
- Mass spectra show the mass to charge (m/z) ratio of ions that are put into it
- The molecular ion will have the highest m/z value with reasonable intensity
- There may be a peak 1 unit higher due to 13C being 1.11% naturally abundant
- This is the M+1 peak, don’t confuse it for the molecular ion
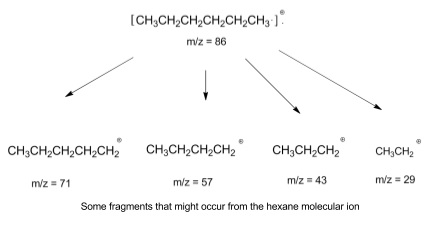
7. Fragmentation patterns
- Excess energy given in forming the molecular ion may cause the molecule to fragment and split into pieces
- Fragmentation can happen anywhere in a molecule, and fragments can break into further fragments
- So lots of different m/z ratios due to different fragments are seen in spectra
- This allows them to be used to identify different molecules
- Two isomers will have the same molecular ion peak, but different fragmentation patterns
- Organic compounds have unique mass spectra, which can be used as a fingerprint for identification
- Two isomers will have the same molecular ion peak, but different fragmentation patterns
- When an ion fragments, one fragment is charged, while the other is neutral
- This can be reversed, so one fragmentation gives two possible fragments that can be detected
- If you get a mass spectrum of an unknown compound it helps to draw out the compound and look at what could fragment off to help explain the m/z peaks
- Table shows some common fragments
m/z value |
Possible fragment ion identity |
15 |
CH3+ |
17 |
OH+ from alcohols |
29 |
C2H5+ |
43 |
C3H7+ |
57 |
C4H9+ |
Exam Tip Different fragments can have the same m/z value, so a peak in a spectrum at 43 from a molecule that you don’t think can fragment to give C3H7+ may well not be that |
8. Finding structures from different analytical data
- Different analytical provide different information so they are used together to work out what compound is being studied
- Elemental analysis gives the percentage of each element in a compound
- Use percentages and Ar values to work out number of moles of each element and empirical/molecular formula
- Mass spectrometry is destructive - the sample can’t be retrieved after analysis
- But it works on very small quantities, is quite cheap and very accurate
- Helps to find relative molecular mass from molecular ion peak
- Fragmentation pattern gives an idea of structure
- IR is non-destructive
- When given data from the three techniques and asked to determine what molecule is present
- Use elemental analysis to find the empirical/molecular formula
- Use mass spec. To find the Mr if not found from elemental analysis
- Also use mass spec. to work out what fragments form
- Use this to find a plausible structure and choose between isomers
- Use IR to determine what functional groups are present and incorporate them into the molecule
- Use elemental analysis to find the empirical/molecular formula
Exam Tip You’ll only need to be able to work out spectra and data relating to compounds with the functional groups covered in this module |