A-LEVEL OCR ChEMISTRY NOTES
Periodicity
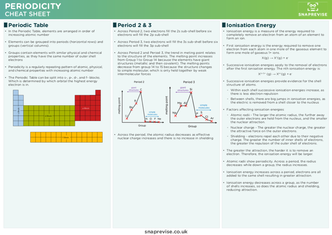
Periodic Table
- In the Periodic Table, elements are arranged in order of increasing atomic number
- Elements can be grouped into periods (horizontal rows) and groups (vertical columns)
- Groups contain elements with similar physical and chemical properties, as they have the same number of outer shell electrons
- Periodicity is a regularly repeating pattern of atomic, physical, and chemical properties with increasing atomic number
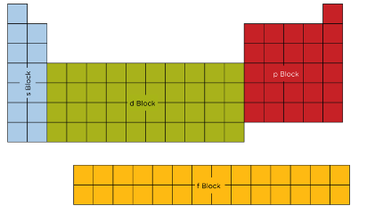
- The Periodic Table can be split into s-, p-, d-, and f- blocks. Which is determined by which orbital the highest energy electron is in
Ionisation Energy
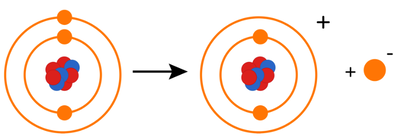
- Ionisation energy is a measure of the energy required to completely remove an electron from an atom of an element to form an ion.
- First ionisation energy is the energy required to remove one electron from each atom in one mole of the gaseous element to form one mole of gaseous 1+ ions.
X (g)→ X+(g) + e-
- Successive ionisation energies apply to the removal of electrons after the first ionisation energy. The nth ionisation energy is:
X^(n-1) + (g)→ X^n+(g) + e^-
- Successive ionisation energies provide evidence for the shell structure of atoms.
- Within each shell successive ionisation energies increase, as there is less electron repulsion
- Between shells, there are big jumps in ionisation energies, as the electron is removed from a shell closer to the nucleus
- Factors affecting ionisation energies:
- Atomic radii- The larger the atomic radius, the further away the outer electrons are held from the nucleus, and the smaller the nuclear attraction.
- Nuclear charge- The greater the nuclear charge, the greater the attractive force on the outer electrons.
- Shielding- electrons repel each other due to their negative charge. The greater the number of inner shells of electrons, the greater the repulsion of the outer shell of electrons.
- The greater the attraction, the harder it is to remove an electron. Therefore, the ionisation energy will be larger.
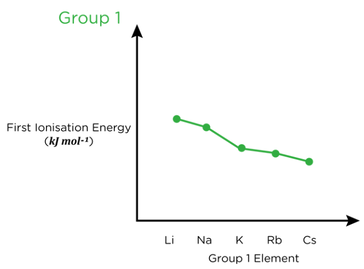
- Atomic radii show periodicity. Across a period, the radius decreases while down a group, the radius increases.
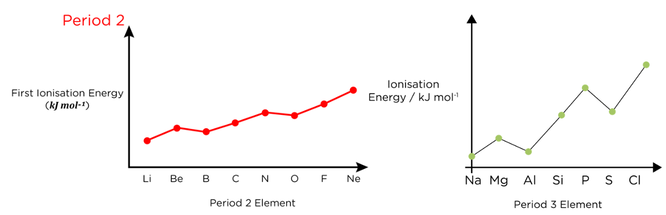
- Ionisation energy increases across a period, as nuclear charge increases, but shielding stays the same
- In Period 3:
- Aluminium is an exception as the 3s electrons shield the 3p electron
- Sulfur is an exception as an electron is being removed from an p orbital containing 2 electrons, so there is electron-pair repulsion
- Ionisation energy decreases across a group, as the number of shells increases, so does the atomic radius and shielding, reducing attraction
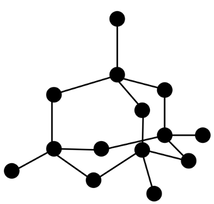
Giant Covalent Structures
- Giant covalent lattices consist of billions of atoms held together by a network of strong covalent bonds.
- Carbon forms three types of giant covalent structures: graphite, graphene and diamond.
- In diamond, each carbon atom is bonded to four other carbon atoms
- This makes it very strong, with an incredibly high melting point, hardness, insolubility. It is an electrical insulator because all the outer shell electrons are being used in covalent bonds, so there are no mobile charge carriers
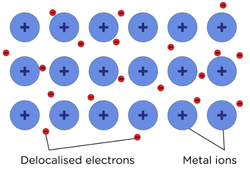
Metallic Bonding
- Metallic bonding is the strong electrostatic attraction between positive metal ions and negative delocalised electrons in a metal lattice
- Metals have a fixed lattice structure of positive ions. The outer shell of electrons is delocalised, which can carry current.
- Metals have high melting and boiling points due to the large amounts of energy needed to overcome the metallic bonds.
- The greater the charge of a metal ion, the more electrons are delocalised. The smaller the size of the metal ions, the closer they are to the delocalised electrons. This results in stronger bonds and a higher melting point
- Metals are malleable and ductile. The ions can slide and move past each other as there are no bonds holding specific ions together
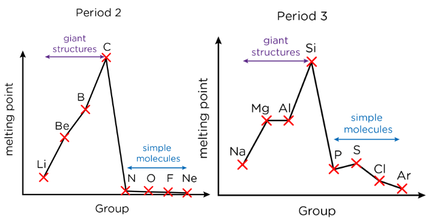
Period 2 & 3
- Across Period 2, two electrons fill the 2s sub-shell before six electrons will fill the 2p sub-shell
- Across Period 3, two electrons will fill the 3s sub-shell before six electrons will fill the 3p sub-shell
- Across Period 2 and Period 3, the trend in meting point relates to the structure of the elements. The melting point increases from Group 1 to Group 14 because the elements have giant structures (metallic and then covalent). The melting points decrease from group 14 to 15 because the structure changes to simple molecular, which is only held together by weak intermolecular forces
- Across the period, the atomic radius decreases as effective nuclear charge increases and there is no increase in shielding