A-LEVEL OCR ChEMISTRY NOTES
Carboxylic Acids and Esters
Carboxylic Acids & Esters
- Carboxylic acids have the functional group -COOH
- Carboxylic acids partially dissociate in water, making them weak acids
- Carboxylic acids can react with carbonates to liberate CO2.
- 2CH3COOH (aq) + Na2CO3 (s)→2CH3COO-Na+ (aq) + CO2 (g) + H2O (l)
- They also react with alkalis and metal oxides as acids do
- Carboxylic acids with short carbon chains (up to four carbons) are soluble because they can form hydrogen bonds with water molecules
- The C=O and O-H bond in carboxylic acids are polar bonds, allowing for the hydrogen bonds to form
- As the chain length increases, the solubility decreases, because the size of the hydrophobic non-polar part of the molecule increases
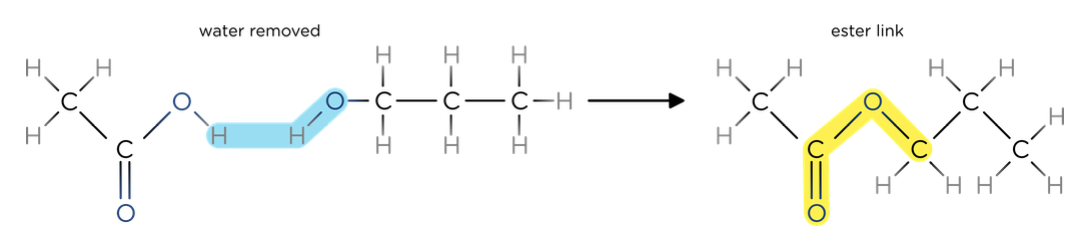
- Esters have the general formula RCOOR’.
- Esters can be made when alcohols and carboxylic acids are reacted in the presence of a sulphuric acid catalyst
- The alcohol gives the first part of the name and the acid gives the second part of the name
- Esters are used as solvents, plasticisers, perfumes and food flavourings
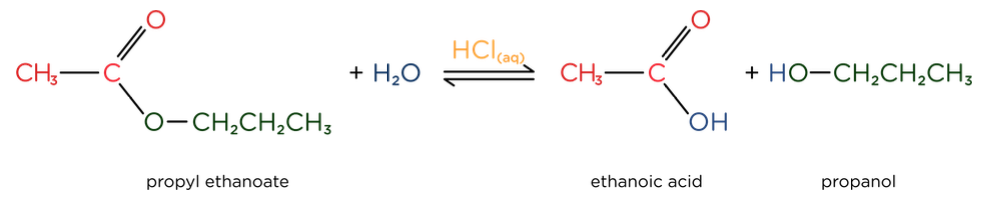
- Acid catalysed hydrolysis of esters forms a carboxylic acid and an alcohol
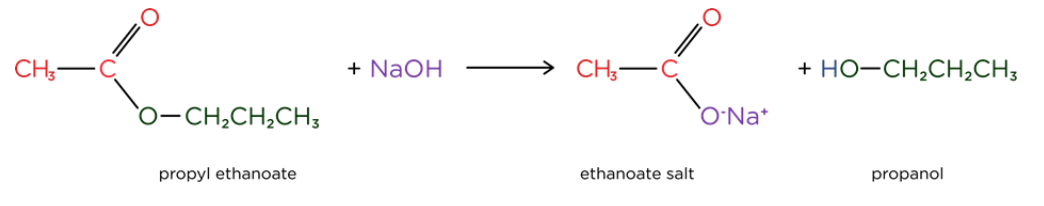
- Base catalysed hydrolysis of ester forms a carboxylate salt and an alcohol
Acylation
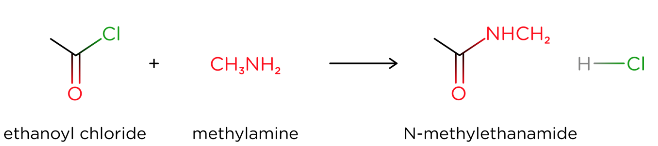
- Acyl chlorides are a derivative of carboxylic acids, where the -OH group is replaced by a -Cl group
- They are prepared by reacting a carboxylic acid with SOCl2:
- CH3COOH + SOCl2 → CH3COCl + SO2 + HCl
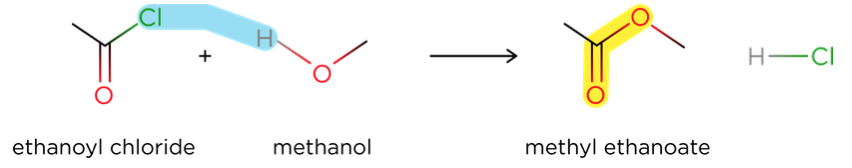
- Acyl chlorides react with alcohols to form esters in a nucleophilic addition-elimination reaction.
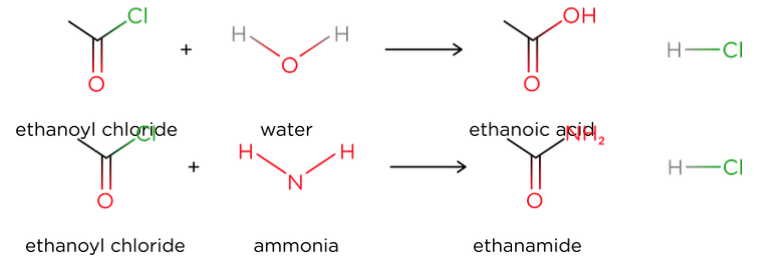
- Acyl chlorides will react with a variety of nucleophiles