A-LEVEL OCR ChEMISTRY NOTES
Carbonyl Compounds
Aldehyde & Ketones
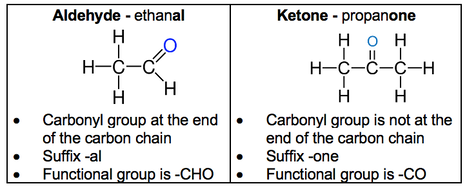
- Aldehydes and ketones are carbonyl compounds containing the C=O functional group.
- Aldehydes can be oxidised to form carboxylic acids by heating under reflux with potassium dichromate (oxidising agent) and concentrated sulphuric acid
- Carbonyls have a permanent dipole making them susceptible to nucleophilic addition reactions.
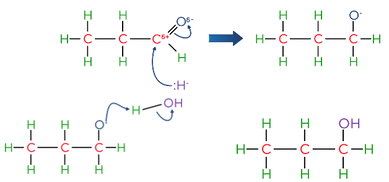
- Aldehydes can be reduced to primary alcohols and ketones to secondary alcohols using NaBH4 as the reducing agent e.g. reduction of propanal CH3CH2CHO + 2[H] → CH3CH2CH2OH
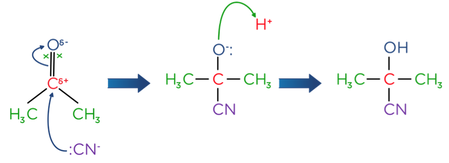
- Nucleophilic addition of cyanide ions can be used to extend the length of a carbon chain
- Reactions of carbonyls with KCN and dilute acid, produces a hydroxynitrile. Aldehydes and unsymmetrical ketones form two optical isomers, as there is an equal chance of either isomer being formed. This is a racemic mixture.
- Cyanide-containing compounds can be toxic or irritants.