A2-LEVEL OCR ChEMISTRY NOTES
TOPIC 3: Transition elements
1. transition elements
1. Electron configurations of period 4 d-block atoms and ions
- Write using sub-shell notation, filling lowest energy sub-shells first
- The 4s sub-shell is filled before the 3d
- Cr and Cu only have one electron in the 4s orbital and 5 and 10 respectively in the 3d orbital
- When forming ions, 4s electrons are lost before 3d
2. Transition elements
- Ti → Cu are transition elements
- They have a stable ion with an incomplete 3d sub-shell
3. Properties of transition elements
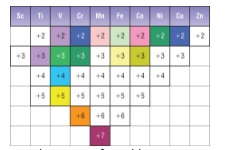
- Variable oxidation states
- Ti → Cu all form 2+ ions (+2 oxidation state)
- They all form at least one other oxidation state
- Ti → Cu all form 2+ ions (+2 oxidation state)
- Coloured ions
- Most Ti → Cu ions are coloured in solution
- Colour comes from ions absorbing a specific colour
- Colour observed is a mix of all colours not absorbed
- Most Ti → Cu ions are coloured in solution
- Catalysts have two main ways of working
- Provide a surface for reactions to happen on
- Provide an alternative reaction pathway with a lower activation energy
- Haber process uses iron metal
- Contact process uses V2O5
- Alkene hydration uses nickel metal
- Decomposition of H2O2 uses MnO2
- Laboratory production of H2 gas uses CuSO4
Exam Tip Don’t learn the properties of all Ti → Cu metals, you only need to know two examples of specific metals for the three properties above i.e. two metals that are catalysts, two coloured ions, two sets of variable oxidation states |
4. Ligands
5. Complex ions
6. Two types of stereoisomerism are found in transition metal compounds
- Molecules or ions that donate a pair of electrons to a metal ion
- Form coordinate/dative covalent bonds
- Examples are H2O, Cl-, NH3
- Can be monodentate (form one bond to the metal) or bidentate (form two bonds to the metal, one each from two separate atoms) e.g. ‘en’
5. Complex ions
- A central metal ion bonded to one or more ligands
- Written in square brackets with charge outside e.g. [Co(H2O)6]2+
- Coordination number is the number of ligands bonded to the metal ion
- Commonly four or six coordinate
- Six coordinate are all octahedral e.g. [Fe(H2O)6]2+, [Ni(en)3]2+
- Four coordinate complexes can be square planar or tetrahedral
- Tetrahedral: tetrachloro complexes e.g. [CoCl4]2+,[CuCl4]2+
- Square planar: Pt complexes e.g. [Pt(Cl2)(NH3)2]2+
- Six coordinate are all octahedral e.g. [Fe(H2O)6]2+, [Ni(en)3]2+
6. Two types of stereoisomerism are found in transition metal compounds
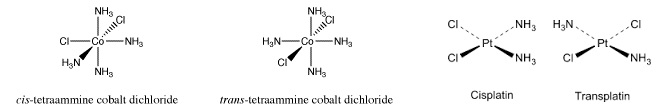
- Cis-trans isomerism
- Found in octahedral/square planar complexes with only two different kinds of ligands
- Octahedral complexes need four of one ligand (or two bidendate) and two of another
- Found in octahedral/square planar complexes with only two different kinds of ligands
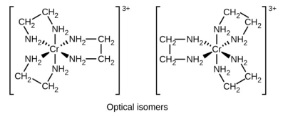
- Optical isomerism
- Non-superimposable mirror images
- Complex must have one of three sets of ligands
- Three of the same bidentate ligand
- Two of the same bidentate ligand and two identical monodentate ligands
- One hexadentate ligand (one ligand forming six bonds)
- Non-superimposable mirror images
7. Cis-platin
- Square planar Pt compound [Pt(Cl2)(NH3)2]2+
- Anti-cancer drug
- Probably acts by binding to cancer cell DNA and preventing cell division
- Involves ligands in complex ions being replaced by other ligands. There are examples involving copper and chromium
- Blue copper hexaaquo, [Cu(H2O)6]2+, reacts with ammonia and HCl
- Adding a small amount of ammonia produces pale blue Cu(OH)2 as a precipitate
- Adding an excess produces [Cu(NH3)4(H2O)2]2+
- Adding HCl produces yellow [CuCl4]2-
- Initially this combines with blue [Cu(H2O)6]2+ to make a green solution
- Eventually this turns yellow with enough HCl and [CuCl4]2- predominates
- Dark green/grey-green [Cr(H2O)6]3+ reacts with ammonia
- Adding a small amount of ammonia produces a green precipitate [Cr(H2O)3(OH)3]
- Adding an excess dissolves some of the precipitate and leads to ligand substitution, forming [Cr(NH3)6]3+
- Red blood cells contain haemoglobin which contains Fe2+
- Oxygen acts as a ligand to bind reversibly to Fe2+ and is carried round the body
- CO binds more strongly to haemoglobin than O2 and is toxic to humans
- If present in high concentrations it binds irreversibly to haemoglobin, reducing the amount that can bind O2
- This makes it hard for the body to get enough oxygen, which can be fatal
- Soluble ions in separate solutions react to form an insoluble precipitate
Ion |
Observation with NaOH |
Observation with NH3 |
Fe2+(aq) |
Pale green Fe2+ solution forms a green precipitate which turns rusty brown at the surface in air as Fe2+ oxidises to Fe3+ Fe2+(aq) + 2OH-(aq) → Fe(OH)2(s) |
Dark green precipitate forms [Fe(H2O)6]2+(aq) + 2NH3(aq) → [Fe(OH)2(H2O)4](s) + 2NH4+(aq) |
Fe3+(aq) |
Pale yellow Fe3+ solution forms a rusty brown precipitate Fe3+(aq) + 3OH-(aq) → Fe(OH)3(s) |
Rusty brown precipitate forms [Fe(H2O)6]3+(aq) + 3NH3(aq) → [Fe(OH)3(H2O)3](s) + 3NH4+(aq) |
Mn2+(aq) |
Brown precipitate forms which darkens as it oxidises in air Mn2+(aq) + 2OH-(aq) → Mn(OH)2(s) Oxidation: 4Mn(OH)2(s) + O2(g) → 2Mn2O3(s) +4H2O(l) |
Brown precipitate forms which darkens as it oxidises in air [Mn(H2O)6]2+(aq) + 2NH3(aq) → [Mn(OH)2(H2O)4](s) + 2NH4+(aq) |
Cu2+(aq) |
Blue, gelatinous precipitate forms Cu2+(aq) + 2OH-(aq) → Cu(OH)2(s) |
Blue, gelatinous precipitate forms which re-dissolves to a blue solution [Cu(H2O)6]2+(aq) + 2NH3(aq) → [Cu(OH)2(H2O)4](s) +2NH4+(aq) Overall: [Cu(H2O)6]2+(aq) +4NH3 → [Cu(NH3)4(H2O)2]2+(aq) +2H2O(l) |
Cr3+(aq) |
Green precipitate forms, which re-dissolves in excess OH- [Cr(H2O)6]3+(aq) + 3OH-(aq) → [Cr(OH-)3(H2O)3](s) +3H2O(l) Then: [Cr(OH-)3(H2O)3](s)+ 3OH-(aq) → [Cr(HO)6]3-(aq) + 3H2O(l) |
Green precipitate forms, which re-dissolves in excess ammonia [Cr(H2O)6]3+(aq) + 3NH3(aq) → [Cr(OH-)3(H2O)3](s) +3NH4+(l) Then: [Cr(OH-)3(H2O)3](s)+ 6NH3(aq) → [Cr(NH3)6]3+(aq) + 3H2O(l) + 3OH-(aq) |
Exam Tip You don’t need to use full complex formulae for precipitation reactions, you can just write the metal ions and what they’re reacting with. Also the only ions which have different reactions with a small amount and an excess of OH-/NH3 are Cu2+ and Cr3+ |
11. Redox reactions of transition elements
- Transition metal elements have variable oxidation states so can be used in redox titrations
- Fe2+/Fe3+
- Fe2+ (green) oxidised by Mn(VII)O4- to Fe3+ (brown)
- Mn(VII)O4- (deep purple) reduces to Mn2+ (very pale pink)
- MnO4- solutions need to be acidified with H2SO4 for titrations
- Fe3+ can then be reduced by I- to Fe2+
- Fe2+ (green) oxidised by Mn(VII)O4- to Fe3+ (brown)
- Cr6+/Cr3+
- Cr(VI) oxidises Fe2+ to form Cr(III)
- Cr(III) can be oxidised by H2O2/OH-
- Cr(VI) oxidises Fe2+ to form Cr(III)
- Cu2+/Cu+
- Cu2+ is reduced by I- to Cu+
- Cu+ can then disproportionate
- One mole of Cu+ splits into half moles of Cu(s) and Cu2+
- Cu2+ is reduced by I- to Cu+
- Work out transition metal titrations in the same way as normal redox titrations
Exam Tip If you’re given a question with redox pairs you don’t know, you’ll still be able to work them out in the same way as normal, just be careful working out the balanced equation from half equations (which you’ll be given) to get the rest of the question right |
2. qualitative analysis
- Tests for ions
- Qualitative tests for ions allow us to work out whether a species is present, but now how much of it there is
- Cations
- Half fill a test-tube with the test solution
- Add reagents drop by drop
- Half fill a test-tube with the test solution
- Qualitative tests for ions allow us to work out whether a species is present, but now how much of it there is
Ion |
Colour in solution |
With NH3(aq) or OH-(aq) |
Excess NH3(aq) |
Excess OH-(aq) |
Cu2+ |
Blue |
Blue precipitate |
Precipitate re-dissolves to a blue solution |
No change |
Cr3+ |
Violet |
Green precipitate |
Precipitate re-dissolves to a purple solution |
Precipitate re-dissolves to a green solution |
Fe2+ |
Green |
Green precipitate |
No change |
No change |
Mn2+ |
Pink |
Brown precipitate |
No change |
No change |
Fe3+ |
Yellow/brown |
Brown precipitate |
No change |
No change |
- Anions
- CO32-
- Reacts with acid to produce a gas (CO2) that turns limewater milky
- SO42-
- Reacts with HCl(aq) and BaCl2(aq) to give a white precipitate (BaSO4)
- Halide ions
- Add dilute nitric acid and aqueous silver nitrate
- CO32-
Halide |
Precipitate with AgNO3(aq) |
Solubility of precipitate |
Cl- |
White |
Soluble in dilute NH3(aq) |
Br- |
Cream |
Soluble in conc. NH3(aq) |
I- |
Yellow |
Insoluble in conc. NH3(aq) |
Exam Tip You need to carry out the tests in this order, otherwise compounds might be formed that could be interpreted as the wrong result |