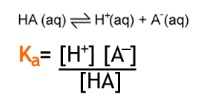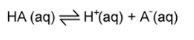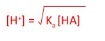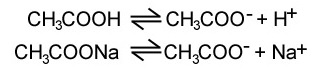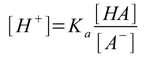A2-LEVEL OCR ChEMISTRY NOTES
TOPIC 1: rates, equilibrium and ph
1. how fast?
1. Key terms
2. Orders and the rate equation
3. Calculating the rate constant, k
- Rate of reaction: the amount of reactant used up, or product that is created, over time
- Normally measured in mol dm-3 s-1, but other units may be used if they are more appropriate, for example if the reaction is very slow we may use mol dm-3 min-1
- Order of reaction: Order with respect to a particular reactant tells you how the reactant’s concentration affects the rate. These can only be found from experiment
- Overall order: the sum of the individual orders for each reactant
- The rate constant, k: a constant of proportionality in the rate equation that can be calculated from the orders and rates of reaction
- Half-life: The time taken for half a reactant to be used up
- Rate-determining step: The slowest step in a multi-step reaction
2. Orders and the rate equation
- We can find the orders of reactants from concentration-time and concentration-rate graphs (more on this later)
- Use the orders of each reactant to create a rate equation of the form rate = k[A]m[B]n
- m and n are the orders of reactants A and B
3. Calculating the rate constant, k
- If we know the rate (mol dm-3 s-1), concentrations of reactants (mol dm-3) and overall order, we can work out the rate constant.
- You can also find its units
Zero order |
Rate = k[A]0 = k |
k = rate/1 |
Units = mol dm-3 s-1 |
First order |
Rate = k[A] |
k = rate/[A] |
Units = s-1 |
Second order |
Rate = k[A]2 |
k = rate/[A]2 |
Units = dm3 mol-1 s-1 |
Third order |
Rate = k[A]2[B] |
k = rate/[A]2[B] |
Units = dm6 mol-2 s-1 |
Exam Tip You’ll need to be able to work out the units for other quantities like equilibrium constants as well, so it’s worth making sure you know how to do it |
4. Concentration-time graphs
- By measuring how the concentration of a reactant changes over time during a reaction we can create a concentration-time graph
- You can use the shape of a concentration-time graph to work out if the reaction is zero or first order (but not if it’s second order)
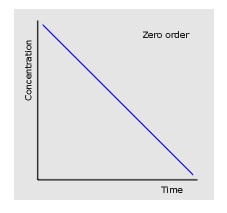
- Zero order: The rate doesn’t change as concentration falls so the graph is a straight line.
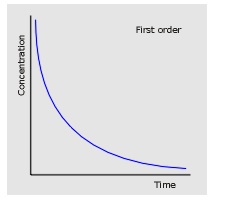
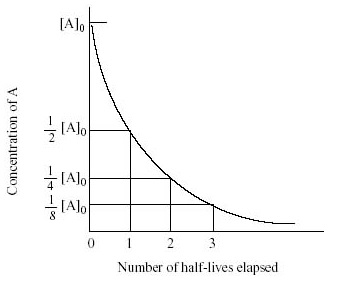
2. First order: The graph has a characteristic curved shape with constant half-life (halving of reactant concentrations at equal time intervals)
- To find the rate at any time take the gradient of the graph
- For zero order this will just be the gradient of the line
- For first order you need to take a tangent to the graph
5. Experimental Techniques to find Rate Data:
- For reactions with acids or bases measure pH changes by doing titrations or using a pH meter
- If a product of the reaction is a gas, measure the volume of gas produced at regular time intervals
- Volume of gas is proportional to moles of gas
- So it can be used to measure the amount of product produced
- Colorimetry is used if the products or reactants are coloured
- Reactants are mixed and timer is started
- Light absorbance at a particular wavelength is measured at set time intervals
- Reactants are mixed and timer is started
6. Half life
- From a concentration-time graph of a first order reaction you can find the half-life from the time it takes the concentration to decrease by a half
7. Half life and the rate constant
8. Clock reactions
9. Rate concentration graphs
- The half life is related to the rate constant by k = ln2/t1/2
8. Clock reactions
- We can use the ‘clock’ reactions like the iodine clock to plot rate-concentration graphs
- Reactants are mixed and time, t, taken to produce a fixed amount of product is measured
- The experiment is repeated using several different starting concentrations
- The shorter the time taken, the faster the reaction must be i.e. rate ∝ 1/t
- We take 1/t as a good approximation of the initial rate, and can use this to plot rate-concentration graphs
9. Rate concentration graphs
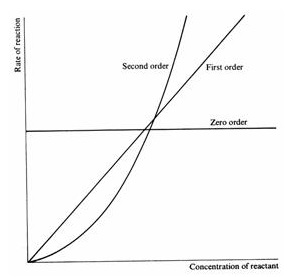
- We can use rate-concentration graphs to determine the order of the reaction with respect to a reactant A
- Zero order, rate ∝ [A]0 - horizontal line. Changes in the concentration of this reactant have no effect on the rate
- First order, rate ∝ [A]1 - straight line. If [A] doubles then the rate will double. Since rate ∝ [A], rate = k[A], i.e. k=rate/concentration and we can find k as the gradient of the graph
- Second order, rate ∝ [A]2 - curved line. If [A] doubles the rate will increase by 22
10. For a multi-step reaction
- The rate determining step will be the slowest step in the reaction mechanism
- The rate equation includes all species that appear in or before the rate determining step
- If all reactants in the overall balanced equation are in the equation, the reaction is single step
- If any reactants are zero order , there must be at least one additional step
- The overall equation includes all species present in the mechanism that aren’t intermediates
- An intermediate is something that is produced and used up during the mechanism
- Reactions occur when particles collide in the correct orientation and with a combined energy above the activation energy
- Increasing the temperature increases the number of collisions, but more importantly the number that occur above the activation energy
- Raising the temperature speeds up most reactions by increasing k
- For many reactions, rate doubles with every increase in temperature of 10oc
- An exponential relationship between k and T
- k = Ae-Ea/RT
- The equation and R are given to you in the databook
- A is a pre-exponential factor, you don’t need to know what it represents but you might need to find it from a graph
- As T increases, k increases
- It can be put into logarithmic form lnk = lnA - Ea/R x 1/T
- This form is similar to the equation of a line
- Plot lnk against 1/T
- This will have a gradient of –Ea/R
- We can then use the gradient to find the activation energy, and the intercept to find A
Exam Tip The Arrhenius equation and R are given to you in the data book so you don’t need to learn them |
2. how far?
1. Mole fractions and partial pressure
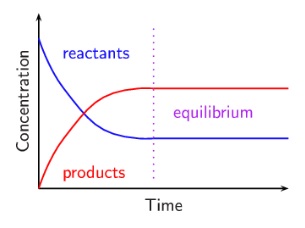
2. Some reactions don’t go all the way to completion, and instead produce a mixture of products and reactants called an equilibrium:
- A mole fraction, XA, of substance, A, is the fraction of the total moles in a system that are of substance A
- The partial pressure, PA, of a gas, A, in a mixture of gases is the pressure it would exert if it was alone in the container
2. Some reactions don’t go all the way to completion, and instead produce a mixture of products and reactants called an equilibrium:
- This is the state at which the rate of the forwards reaction is equal to the backwards reaction
- Concentration of products and reactants remain the same
- A homogeneous equilibrium involves reactants and products in the same state (e.g. solid, liquid, gas)
- A heterogeneous equilibrium exists if more than one state is present
3. Finding concentrations at equilibrium
- Two main ways
- Titration
- Titrate one of the reactants or products against a solution of known concentration to find its concentration
- Find the concentrations of other species from the balanced equation
- Colorimetry
- A colorimeter measures the concentration of one species throughout the reaction
- Find the concentrations of other species from the balanced equation
- At equilibrium, the concentrations of the products and reactants are related by an equilibrium constant, Kc
- For the reaction mA + nB ⇋ pC + qD:
- Kc= [C]p[D]q[A]m[B]n
- Work out the units of Kc in the same way as the units for the rate constant, by cancelling out the units of each separate term
- When a system involving gases reaches equilibrium, a relationship between the partial pressures of the products and reactants is established
- The partial pressures are related by a different (but really similar) constant Kp
- For the reaction mA + nB ⇋ pC + qD where all species are gases:
- Kp= (PC)p(PD)q(PA)m(PB)n
- Solids and pure liquids are not included in the expressions for Kc and Kp
- If K is bigger than 1, there will be more products at equilibrium, if K is less than 1, the reactants will predominate
Exam Tip In equilibrium questions you’ll often be given a chemical equation and some information about the initial number of moles of reactants and the number of moles of products formed. It helps to make a table with this info to work out the equilibrium number of moles of each substance. |
5. Kc, Kp and temperature
- Kc and Kp are only affected by temperature
- They aren’t affected by changing concentrations, pressures or catalysts
- In an endothermic reaction, an increase in temperature causes an increase in K
3. acids, bases and buffers
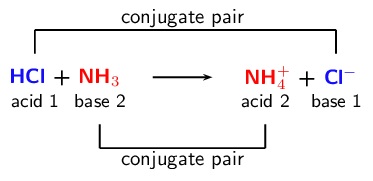
1. Bronsted-Lowry acids and bases
- In the Bronsted-Lowry theory of acids and bases
- Acids are substances that donate protons to a base
- Bases are substances that accept protons from an acid
- Acids are substances that donate protons to a base
- Conjugate Pairs
- Acids form their conjugate bases through the loss of a proton
- Bases form their conjugate acids through the gain of a proton
- Acids form their conjugate bases through the loss of a proton
- Acids can be monobasic, dibasic or tribasic
- Monobasic acids produce 1 mole of H+ per mole of acid
- Dibasic acids produce 2 moles of H+ per mole of acid
- Monobasic acids produce 1 mole of H+ per mole of acid
2. Reactions of acids
- The H+ released by acids can react with metals and undergo neutralisation reactions with bases such as carbonates, metal oxides and alkalis.
Word Equation |
Chemical Equation |
Ionic Equation |
acid + carbonate → salt + water + CO2 |
2HCl(aq) + CaCO3(s) → CaCl2(aq) + H2O(l) + CO2(g |
2H+(aq) + CO32-(aq) → H2O(l) + CO2(g) |
acid + metal oxide → salt + water |
2HCl(aq) + MgO(s) → MgCl2(aq) + H2O(l) |
2H+(aq) + MgO(s) → H2O(l) + Mg2+(aq) |
acid + alkali → salt + water |
HNO3(aq) + NaOH(aq) → H2O(l) + NaNO3(aq) |
H+(aq) + OH-(aq) → H2O(l) |
acid + metal → salt + hydrogen |
2HCl(aq) + Zn(s) → ZnCl2(aq) + H2(g) |
H+(aq) + Zn(s) → H2(g) + Zn2+(aq) |
Exam Tip You’re only expected to be able to write the ionic equations for these reactions. Ionic equations leave out all the spectator ions in a reaction |
3. The acid dissociation constant, Ka
- Shows how dissociated an acid is in solution
- Ka has units of mol dm-3
- Strong acids completely dissociate in solution and have large Ka values
- Weak acids only partially dissociate in solution and have small Ka values
Exam Tip Don’t confuse how strong and weak acids and bases dissociate with their concentrations. You can have low concentrations of strong acids and high concentrations of weak acids |
4. pH
- Similarly to Ka, a huge range of acidities exist
- We use pH as a measure of the acidity (specifically the concentration of H+(aq) ions) of a solution.
5. The ionic product of water
6. Finding the pH of strong acids and bases
7. Finding the pH of weak monobasic acids
- Water itself dissociates to a small extent through H2O(l) → H+(aq) + OH-(aq).
- Kw, the ionic product of water, shows how much dissociation has occurred
- Kw = [OH-(aq)][H+(aq)]
- Water is pH 7 at 25oC, so [H+(aq)] = 1x10-7 mol dm-3
- Pure water produces the same number of OH- and H+ ions when it dissociates
- So at 25oC, Kw is 1x10-14
6. Finding the pH of strong acids and bases
- Strong acids
- Strong acids are fully dissociated so we can assume that [HA(aq)] =[H+(aq)]
- The pH can then be found using pH = -log10[H+(aq)]
- Strong bases
- We use the fact that Kw at 25oC is always 1x10-14
- Strong bases also fully dissociate, so in the equation above [OH-(aq)] = [NaOH(aq)], but this works for any strong base
- Then we can use [H+(aq)] = Kw/[OH-(aq)] and use [H+(aq)] to find pH
7. Finding the pH of weak monobasic acids
- These are only partially disassociated
- Bases/acids are in equilibrium with their conjugate acids/bases
- We assume that [H+] =[A-] and [HAinitial] = [HAequilibrium]
8. The limitations of assumptions
- The assumptions for weak acids get less accurate the more an acid dissociates
- If more than about 5% of the acid dissociates, the assumption that [HAinitial] = [HAequilibrium] can’t be used
- Resist changes to pH when small amounts of acid or base are added
- Buffer solutions can be made from a mixture of a weak acid and a salt of its conjugate base pair, e.g. CH3COOH and CH3COONa
- The sodium ethanoate completely dissociates, giving a reservoir of CH3COO-
- If H+ is added to the solution, it reacts with CH3COO-, pushing the first equilibrium to the left and minimising pH change
- If OH- is added, it reacts with CH3COOH, pushing the first equilibrium to the right and minimising pH change
- They can also be made from a mixture of a weak acid and a strong alkali
11. Finding the pH of buffer solutions
- The pH of a buffer solution can be found from the Ka of the weak acid and the concentration ratio of the weak acid and its conjugate base
- The equation for Ka rearranges to
- We assume that much more A- is from the salt than from HA, so [A-] = [Salt]
- We also assume HA only weakly dissociates, so that [HA] = [Weak Acid]
- You’ll normally be given Ka, so pH can then be found from [H+]
12. Blood is a buffer system
- Blood plasma maintains a pH of 7.4
- This is due to a mixture of carbonic acid and hydrogen carbonate ions
- Levels of carbonic acid are maintained by an enzyme that converts it to CO2 which can be exhaled
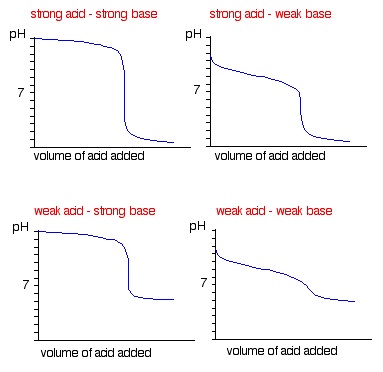
13. Titration Curves
- Show how the pH of a solution varies as the reagent is added
- Let us find the equivalence point, where one solution has reacted exactly with another solution
- It is halfway up the vertical section of the curve
- Shape depends on the strength/weakness of the acid and base
- Suitable indicators must have a colour change within the vertical region of the curve
- Indicators can also be understood as weak acids in equilibrium:
- HIn ⇋ H+ + In-
Titration |
Suitable Indicator |
Strong acid/strong base |
Methyl orange, phenolphthalein |
Strong acid/weak base |
Phenolphthalein |
Weak acid/strong base |
Methyl orange |
Weak acid/weak base |
None appropriate |
14. Using a pH meter
- Much more accurate than universal indicator
- The probe has to be calibrated
- Remove it from the storage solution and rinse with deionised water
- Blot dry then place in a solution of known pH
- Repeat with other solutions of known pH
- Remove it from the storage solution and rinse with deionised water