A2-LEVEL OCR ChEMISTRY NOTES
TOPIC 2: Energy
1. lattice enthalpy
1. Lattice enthalpy
- The lattice enthalpy, ΔLEH⦵, is the enthalpy change when one mole of a solid ionic lattice forms from its gaseous ions under standard conditions
- Depends on strength of the forces acting on ions in a lattice
- Strength of forces depends on:
- Product of the charges on ions (larger charges = larger forces)
- Sum of the radii of the cation and the anion (larger radii = smaller forces)
- Extent of covalency (more covalent = smaller forces)
- Product of the charges on ions (larger charges = larger forces)
- Lattice energy decreases down periodic groups
Exam Tip Remember the superscript circle with a line through it means “under standard conditions”, which are 100kPa and 298K |
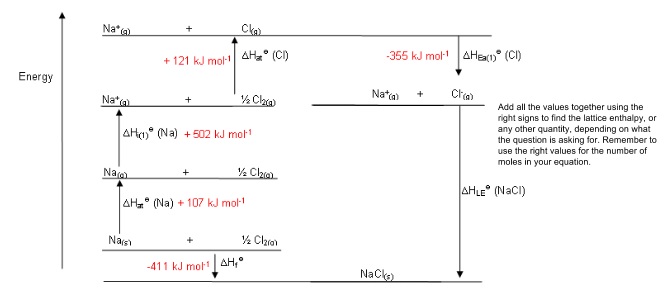
2. Born-Haber cycles
- Lattice enthalpies can be found using Born-Haber cycles,
- These are special versions of Hess cycles
- You can find ΔLEH⦵ if you know the other energy terms in the cycle:
- Enthalpy change of formation, ΔfH⦵: the enthalpy change when 1 mole of a compound is formed from its elements
- (First/Second) Ionisation energy, ΔI(1/2)H⦵: the energy required to remove 1 mole of electrons from a mole of gaseous atoms
- Enthalpy change of atomisation, ΔaH⦵: energy required to turn a molecule of an element into atoms in the gas phase e.g. Cl2(g) → 2Cl(g)
- (First/Second) Electron affinity, ΔEA(1/2)H⦵
- If you need to construct the Born-Haber cycle, start with a line for the elements in their standard states then add each step one by one
Exam Tip You only need to know the definitions of enthalpy change of formation and ionisation energy |
3. Enthalpy changes of solution and hydration
- When an ionic solid dissolves there is a change in enthalpy:
- The enthalpy change of solution, ∆solH⦵, is the enthalpy change when 1 mole of solute is completely dissolved in water
- This can be endothermic or exothermic
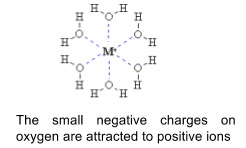
- Positive ions are attracted to the oxygen in water
- Negative ions are attracted to the hydrogen
- The enthalpy change of hydration of an ion, ∆hydH⦵, is related to ∆solH⦵
- It is the enthalpy change when 1 mole of gaseous ions is dissolved in water
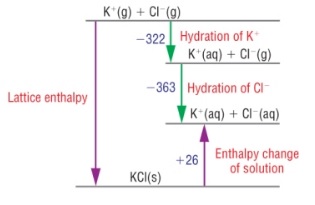
4. Lattice enthalpies from ∆hydH⦵ and∆solH⦵
- ∆hydH⦵ and ∆solH⦵ can be used to make a different Born-Haber cycle to find lattice enthalpies.
- They’re made in exactly the same way as before but using the energy terms:
- Lattice enthalpy
- Enthalpy change of solution
- Enthalpy change of hydration
5. Factors affecting ∆hydH⦵
- The size of the enthalpy of hydration increases with the strength of force between the ion and the water molecules surrounding it,
- This depends on:
- Magnitude of charge of ion (larger charge = stronger force)
- Radius of ion (smaller radius = stronger force)
- Magnitude of charge of ion (larger charge = stronger force)
Exam Tip At A level you can think of energy and enthalpy as being pretty much exactly the same thing |
2. enthalpy and entropy
1. Entropy
2. Entropy of different states of matter
3. Finding the change in entropy, ∆S
4. Spontaneous reactions
- Measure of the disorder of a system, more disorder means higher entropy
- Related to the number of ways of arranging particles
- Particles can be arranged in different ways and have different amounts of energy
- Entropy always tends to increase
- Is often given in units of J K-1
2. Entropy of different states of matter
- Different states of matter have different amounts of entropy because they are more or less ordered
- Entropy of solids < Entropy of liquids < Entropy of gases
- Because gases have the highest entropy, an increase in the number of gas molecules → an increase in entropy, and vice versa for a decrease
3. Finding the change in entropy, ∆S
- ∆S = ΣS⦵products - ΣS⦵reactants
- If ∆S is positive the system has become more random and vice versa
4. Spontaneous reactions
- Whether or not a reaction is feasible depends on the entropy change and temperature of a system, T∆S, as well as the enthalpy change, ∆H
- These are related to the free energy change, ∆G
- ∆G = ∆H - T∆S
- If a reaction happens by itself without having to do anything to it it is spontaneous
- A reaction will be spontaneous only if ∆G < 0
- For large negative ∆H (very exothermic reactions), usually ∆G < 0
- For large positive ∆S (reactions that produce a lot of gas), usually ∆G < 0
ΔH |
ΔS |
ΔG |
Spontaneous |
Negative |
Positive |
Always negative |
Yes |
Positive |
Negative |
Always positive |
No |
Negative |
Negative |
Negative at low T |
At low T |
Positive |
Positive |
Negative at high T |
At high T |
Exam Tip ∆S is often given in JK-1 while ∆H is often in kJ, so you need to remember to convert them if you’re doing a calculation |
6. Kinetic limitations on spontaneous reactions
- Some reactions have ∆G < 0 but don’t happen spontaneously
- There are two kinetic reasons why this might be
- The reaction might have a high activation energy
- The reaction might be extremely slow
- The reaction might have a high activation energy
3. redox and electrode potentials
1. Redox reactions
2. Half Reactions
3. Changes in oxidation numbers can be used to create complete redox reactions
- A redox reaction involves one species being oxidised and another reduced
- Oxidation is carried out by an oxidising agent
- Oxidising agents readily accept electrons and are reduced
- Reduction is done by a reducing agent
- Reducing agents readily donate electrons and are oxidised
2. Half Reactions
- Show either the reduction or the oxidation happening in a redox reaction
- Have electrons on either left hand side (reduction) or right hand side (oxidation)
- Do not include spectator ions
- Can be combined to make full equations
- Balancing the number of electrons in each reaction
- Add the two half equations together and cancel the electrons
- Balancing the number of electrons in each reaction
3. Changes in oxidation numbers can be used to create complete redox reactions
- The sum of all the changes in oxidation number should be zero
- Work out the change in oxidation number for each species
- Balance them by multiplying up numbers of moles of one or more of the species
Exam Tip You can use the reaction conditions to help work out what to add if something is missing from the equation. If they’re acidic, you probably need to add H+, alkaline usually means OH-, and if there’s excess H and O then it’s probably H2O |
4. Redox titrations
- Titrate a known solution of oxidising agent into a reducing agent to work out the concentration of the reducing agent, or the oher way round
- Two key systems, Fe2+/MnO4- and I2/S2O32-
- Fe2+/MnO4-: Mn(VII) in purple MnO4- reduces in the presence of H+
- MnO4-(aq) + 8H+(aq) → Mn2+(aq) + 4H2O(l)
- Overall MnO4(aq) + 8H+(aq) 5Fe2+(aq) → Mn2+(aq) + 4H2O(l) 5Fe3+(aq)
- End point is when a pale pink colour appears due to unreacted MnO4-
- I2/S2O32-: I2 is reduced in the presence of thiosulfate (S2O32-)
- I2 is blue-black in presence of starch, but I- is colourless
- Overall 2S2O32-(aq) + I2(aq) → 2I-(aq) + S4O62-(aq)
Exam Tip You’ll need to know how to describe the experimental procedure needed to carry out one of these titrations |
5. Redox titration calculations
6. Standard Electrode Potential, E⦵
- Redox titrations are used to work out the number of moles/concentration of an unknown reactant
- Work out the number of moles of reactants with given concentrations/volumes
- Work out the balanced equations
- Use them to work out the relationship between moles of species you know and those you’re trying to find
- Use the same process if a question has an unfamiliar redox reagent, you just need to work out a new balanced equation
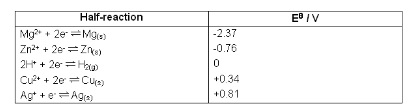
6. Standard Electrode Potential, E⦵
- The electromotive force, emf, of a particular half-cell relative to a standard hydrogen half cell under standard conditions
- A half-cell is a redox half reaction that’s part of an electrical cell
- Always written with the electrons on the left
- E.g. Cu2+ + 2e- ⇋ Cu(s)
- Different half-cells have different tendencies to give/receive electrons so have different e.m.f’s
- Two half cells create a circuit and electrons can flow
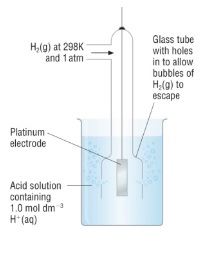
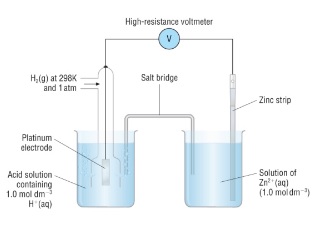
- The standard hydrogen half cell contains three things:
- Inert platinum electrode to allow electrons to flow into/out of the cell
- 1M HCl
7. Measuring half cell potentials
- Metals/non-metals in contact with their ions in aqueous solution
- Connect the half cell to the standard hydrogen electrode
- Link them with a salt bridge and a high-resistance voltmeter
- Read the half cell potential off the voltmeter
- A wire allows electrons to flow
- The salt bridge allows ions to flow between the two solutions
- Connect the half cell to the standard hydrogen electrode
- Measure half cell potentials for ions of the same element in different oxidation states in contact with a Pt electrode in the same way
- Half-cells are arranged in the electrochemical series
- E⦵<0v means the equilibrium lies to the left
- E⦵>0v means the equilibrium lies to the right
Exam Tip You don’t need to know the bit of the electrochemical series above, and the whole thing is huge, you just need to be aware of it and how it’s laid out |
8. Calculating standard cell potentials from standard electrode potentials
9. Predicting the feasibility of reactions from standard cell potentials
10. Fuel cells
11. Electricity generation using fuel cells
- Combine two half-cells with different E⦵ and a current will flow
- The half cell with the more negative (or just less positive) E⦵ undergoes the oxidation reaction (right to left) and vice versa
- Subtract the more negative (or just less positive) E⦵ from the more positive one E⦵ to find the standard cell potential
- Balance the electrons of the right to left (oxidation) reaction of the more negative half-cell and the left to right (reduction) reaction of the more positive half-cell
- Combine the two half-reactions to get the full chemical equation
- Don’t include the electrons in the full equation, cancel them from both sides
9. Predicting the feasibility of reactions from standard cell potentials
- Any species on the right hand side of a half-cell in the electrochemical series will react with any species below it and on the left hand side
- So H2(g) would react with Cu2+
- Reactions that are feasible according to the electrochemical series may not happen for two reasons:
- The concentrations of reactants involved are changed
- This changes the value of E⦵ for a half-cell
- This changes the value of E⦵ for a half-cell
- The rate of reaction might be very slow
- This could be due to a high activation energy
- This could be due to a high activation energy
10. Fuel cells
- One kind of electrochemical cell that’s used to store energy is a fuel cell
- A fuel half-cell and an oxidant half-cell are combined
- The fuel half-cell is selected to have a high energy density
- Work out which is the oxidation/reduction reaction and the cell potential as normal
11. Electricity generation using fuel cells
- The cell generates electricity from the fuel and oxidant
- The fuel and oxidant are consumed to generate a voltage
- The oxidation reaction happens at the anode and the reduction at the cathode