A2-LEVEL OCR ChEMISTRY NOTES
TOPIC 2: nitrogen compounds, polymers and synthesis
1. amines
1. Amines
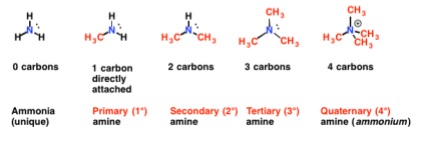
- Organic compounds where one or more of the hydrogens of ammonia has been replaced by an alkyl chain
- To name primary amines:
- Use the carbon chain as the stem
- The suffix is -amine e.g. propylamine
- Add prefixes of any other groups
- Use the carbon chain as the stem
- Secondary amines
- Use the longest carbon chain as the stem
- Add the -amine suffix
- Add the other carbon chain as a prefix
- This is often called an N-substituted amine so begin the name with N-
- Use the longest carbon chain as the stem
- Tertiary amines
- Longest carbon chain as stem
- Add the other two carbons as prefixes
- If there’s more than one of the same chain use di- or tri-
- Begin the name with N-, N-
- e.g. N-, N-dipropylbutamine
- Longest carbon chain as stem
2. Basicity of amines
- Amines and ammonia have a lone pair to donate so are Lewis bases
- They can also accept protons so are Brønsted-Lowry bases as well
- Primary/secondary/tertiary amines react with inorganic acids e.g. HCl to form alkylammonium salts
- RNH2 + HCl → RNH3+Cl-
- RNH2 + HNO3 → RNH3+NO3-
- 2RNH2 + H2SO4 → 2[RNH3+]SO42-
- Replace the hydrogen in the acid with the protonated amine to form the salt
3. Preparation of amines
- Aliphatic amines are made by heating a haloalkane, ammonia and ethanol in a sealed tube as ammonia would escape out of a reflux condenser
- Two stage process
- Haloalkane reacts with ammonia to form ammonium salt
- RCH2X + NH3 → RCH2NH3Cl
- Ammonium salt reacts reversibly with another ammonia molecule
- Forms amine and ammonium chloride
- RCH2NH3Cl + NH3 ⇌ RCH2NH2 +NH4+Cl-
- Excess ammonia drives reaction to the right to from product
- Haloalkane reacts with ammonia to form ammonium salt
- Hydrogen atoms on nitrogen in the amine can be substituted further by the initial haloalkane
- RCH2NH2 + RCH2X ⇌ (RCH2)2NH + HCl
- (RCH2)2NH + RCH2X ⇌ (RCH2)3N + HCl
- (RCH2)3NH + RCH2X ⇌ (RCH2)4N+Cl-
- Final product is known as a quaternary ammonium salt
- A mixture of products will always be formed
- Excess ammonia favours the monosubstituted product
- Excess haloalkane favours the quaternary ammonium salt
- Aromatic amines can be made by reducing nitroalkanes
- Reflux nitroalkane for 30 mins at 100oc with a mixture of tin and conc. HCl
- Add a strong alkali to neutralise the remaining acid and the protonated amine
2. amino acids, amides and chirality
1. Amino acids
- -COOH and -NH2 functional groups attached to the same carbon atom
- General formula RCH2(NH2)COOH
- Proteins are made up of chains of amino acids
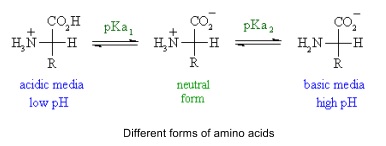
- Carboxylic acid is acidic and amine is basic so amino acids are amphoteric
- React with both acids and bases
- Can form zwitterions where the two groups exchange a proton to from internal salt
- Isoelectric point is where there is no net charge because each amino acid has a balance of charge (as likely to be protonated as deprotonated)
- At low pH lots of amine is protonated
- At high pH lots of carboxylic acid is deprotonated
2. Reactions of amino acids
- Carboxylic acid group undergoes all common reactions with bases
- Metal oxides to neutralise and form salt + water
- Alkalis to neutralise and form a salt + water
- Carbonates to neutralise and form a salt + water + CO2
- Metal oxides to neutralise and form salt + water
- Also react with alcohols to dehydrate (release H2O) and form esters
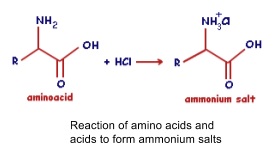
- Amine acts as a base and can react with acids to form ammonium salts
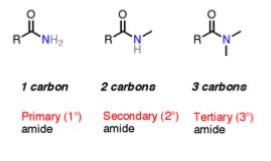
3. Structure of amides
- RC(O)- (acyl group) attached to -NH2
- Can be primary, secondary or tertiary
- Can also be polyamides
- These are condensation polymers made by the reaction of amines and carboxylic acids
- To name primary amides
- Find the stem from the carbon chain attached to the amide
- Suffix is -amide
- Find the stem from the carbon chain attached to the amide
- To name secondary amides
- Stem is from the carbon chain attached to the carbon of the amide
- Suffix is -amide
- Prefix is the alkyl chain attached to the nitrogen
- Alkyl chain is substituted on N so it’s called an N-substituted amide
- e.g. N-ethylpropylamide
- Stem is from the carbon chain attached to the carbon of the amide
4. Optical isomerism
- Example of stereoisomerism
- Molecules that have optical isomers do not have a plane of symmetry
- They are chiral
- Chiral molecules are mirror images of each other
- They are non-superimposable
- These are enantiomers
- Chiral molecules are mirror images of each other
- The most common cause of chirality is a chiral carbon with 4 different groups
- Enantiomers have identical chemical properties
- They differ in only two ways:
- They have different biochemical reactions
- They rotate the plane of polarization of plane polarized light differently
- Enantiomers that rotate plane clockwise have the prefix ‘+’
- Enantiomers that rotate plane anticlockwise have the prefix ‘-’
- They have different biochemical reactions
- 50:50 mixture is called a racemic mixture/racemate
Exam Tip You may need to find chiral centres in a molecule in an exam. This is easy. Just look for carbon atoms with four different groups attached. |
3. polyesters and polyamides
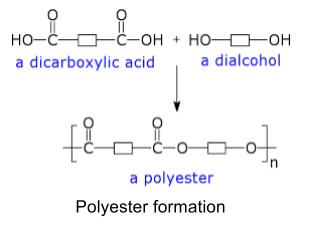
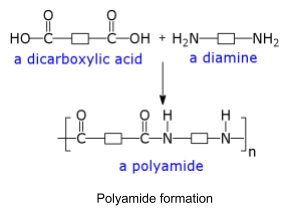
1. Condensation polymerisation
- Happens when two monomers with different functional groups react to form a polymer and release a small molecule in doing so
- One monomer can have both functional groups
- Repeat unit is one monomer if it has both groups
- Repeat unit has one of each monomer if there’s more than one
- Polyesters are formed form monomers that have carboxylic acid and alcohol functional groups (i.e. forming lots of ester bonds)
- Polyamides are formed from monomers that have carboxylic acid and amine functional groups (forming lots of amide bonds)
- This includes amino acids
Exam Tip If a monomer has a C=C bond it forms an addition polymer. If a monomer has two different groups or there’s more than one monomer then a condensation polymer will form |
2. Hydrolysis of polyesters and polyamides
- Can happen at neutral pH but happens very slowly
- Reaction happens faster in acid or base and can be sped up by heating
- Polyesters react slowly in strong aqueous acid to reform the carboxylic acid and alcohol groups of the monomers
- Polyesters react quickly with hot aqueous NaOH to reform the alcohol groups of the monomers and form the salt of the carboxylic acid
- Polyamides react quickly in strong aqueous acid to form the carboxylic acid groups of the monomers and a diammonium salt
4. carbon-carbon bond formation
1. Using the C-C bond to increase the length of the carbon chain
2. Formation of C-CN bonds
- Useful in synthesis when the molecule needs to be made one carbon longer
- -CN is known as a nitrile group
- Can be reacted with organic compounds to add one carbon atom
2. Formation of C-CN bonds
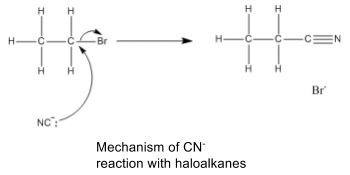
- CN- is a nucleophile, formed from HCN or KCN in a synthesis
- Attacks haloalkanes in a nucleophilic substitution when heated under reflux in an ethanol solvent
- Water not used as a solvent as it would act as the nucleophile instead
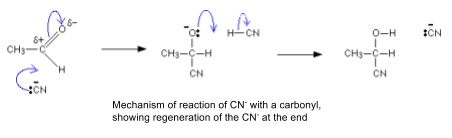
- CN- will also attack the δ+ C of a carbonyl
- This breaks the C=O π bond and the pair of electrons goes to the oxygen atom
- The negative charge on oxygen can then react with H+ to form an OH group
- C=O is planar so CN- can attack from either side to form either stereoisomer if the carbonyl carbon
3. Reaction of nitriles
- Once a nitrile has been formed by one of the reactions above, it can be reacted to form other useful functional groups
- Nitriles can be reduced using a transition metal catalyst or LiAlH4 to form amines
- Nitrile reduced with transition metal catalyst e.g. nickel in the presence of H2 gas heated to 150oc at raised pressure
- RCN + 2H2 → RCH2NH2
- Nitrile reduced by LiAlH4 in ambient conditions
- Reducing agent often abbreviated to [H]
- RCN + 4[H] → RCH2NH2
- Nitriles can be hydrolysed under strong acidic conditions to form carboxylic acids
Exam Tip You don’t need to know the mechanism for this reaction |
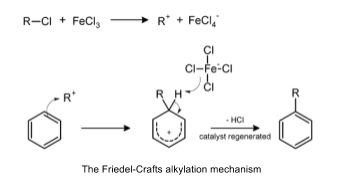
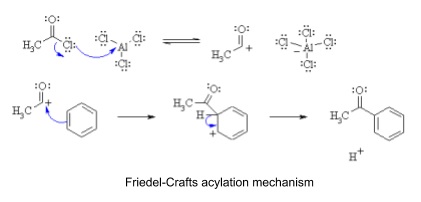
4. Friedel-Crafts reaction
- Friedel-Crafts reaction is electrophilic substitution of an alkyl or acyl group (R+) onto a benzene ring
- Haloalkane and a halogen carrier e.g. FeX3
- R-X + FeX3 → R+ + FeX4-
- Room temperature
- Multiple substitutions likely because alkyl group donates electrons into the delocalised π system
- Makes it more electrophilic
- Products can be separated by fractional distillation/chromatography
- Acyl chloride can also be used
- Very reactive, but refluxed at 60oc for 30 minutes in anhydrous conditions
- Carbonyl group withdraws electrons from aromatic ring so make it less electrophilic
- Less reactive so only one substitution happens
5. organic synthesis
1. Organic preparation
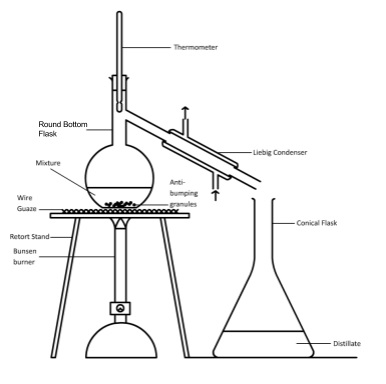
- Quickfit apparatus used in practical organic chemistry
- Grease joints with petroleum jelly to get a good fit or use plastic connectors
- Distillation is a separating technique used to collect product from an organic synthesis reaction
- Set up using clamp stands and bosses to hold equipment
- Connect bottom of condenser to tap with rubber hose and run rubber hose from top of condenser to sink and turn on gentle flow of water
- Pour reaction mixture into flask with anti bumping granules
- Heat and note temperature that products starts evaporating and being collected
- This is its boiling point and helps to identify it
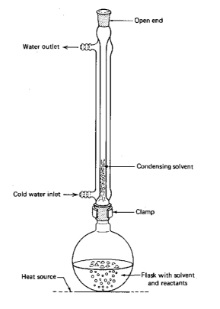
- Heating under reflux is a technique used to ensure that volatile compounds in a reaction mixture don’t boil away
- Several different methods of purifying organic solids
- Organic solids may crystallise during a synthesis, they can be separated from the liquid they crystallised from using filtration under reduced pressure
- Connect rubber tubing to vacuum pump and check there is suction
- Put a Buchner funnel into a clamped filter flask (or a Hirsch funnel if using small quantities)
- Connect tubing from pump to side arm of the filter flask and start suction
- Put filter paper in funner
- Use solvent to dampen the paper so it sticks
- Pour reaction mixture into funnel
- Wash reaction vessel with solvent and add to funnel
- Wash the collected solid with more solvent and maintain suction for a minute after
- Turn off suction and collect solid in watch glass
- Recrystallisation is a technique used to purify product from unreacted starting materials, catalysts and side products
- Three steps
- Crude product dissolved in minimum amount of hot solvent, activated charcoal can be added to remove coloured impurities
- Hot solution quickly gravity filtered in fluted filter paper and filtrate allowed to cool. If no crystals form scratch side of conical flask with glass rod or crash cool in ice bath
- Purified crystals can be separated using vacuum filtration, washes can be done with cold solvent to remove filtrate containing dissolved impurities
- Crude product dissolved in minimum amount of hot solvent, activated charcoal can be added to remove coloured impurities
- Melting points can be used to determine purity
- Every compound has a specific melting point or melting point range
- Impurities will alter this
- Two methods to determine melting point
- Melting point apparatus
- Put a few grains of solid in a capillary tube, insert into apparatus along with accurate thermometer
- Switch on machine with heating dial on ~4
- Look through lens and note the temperatures for when they begin to melt and are fully liquefied
- This is the melting point range
- Put a few grains of solid in a capillary tube, insert into apparatus along with accurate thermometer
- Thiel tube
- Put a few grains of solid in a capillary tube, attach to a thermometer with a rubber band and submerge in the oil of the tube, with rubber band above the oil line
- Heat the side arm with a gentle flame, note the temperature range for the solid starting and finishing melting
- Put a few grains of solid in a capillary tube, attach to a thermometer with a rubber band and submerge in the oil of the tube, with rubber band above the oil line
2. Compounds with multiple functional groups
- It helps to circle functional groups to identify them and make sure you don’t miss complex ones like amides
- Use the properties and reactions of compounds with the individual functional groups to suggest properties and reactions for a compound with multiple groups
Functional groups |
Reactions |
Unsaturated compound (C=C bond) |
|
Haloalkanes |
|
Carbonyl |
|
Carboxylic acids |
|
Alcohol |
|
Esters |
|
3. Multi-stage synthetic routes
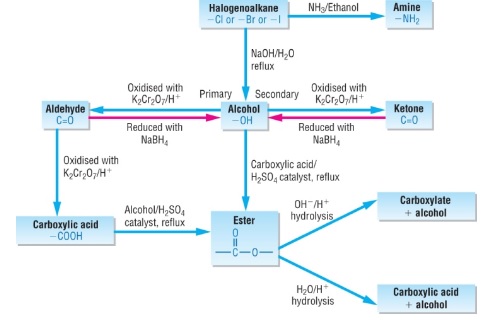
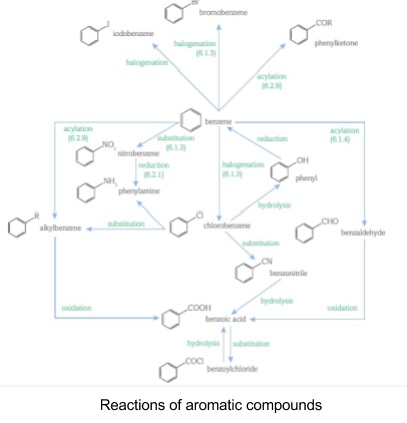
- Need to be able to suggest four-step reactions to get from one compound to another
- Aliphatic reactions are summarised in the flow chart below
- Aromatic reactions are summarised in the flow chart below