A2-LEVEL OCR ChEMISTRY NOTES
TOPIC 1: aromatic compounds, carbonyls and acids
1. aromatic compounds
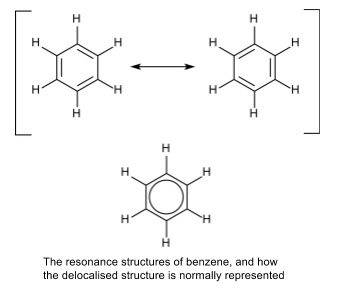
1. The Kekulé model of benzene
- Benzene’s structure was initially suggested by Kekulé to be a ring with alternate double and single bonds
- The actual structure is a resonance structure which is a hybrid of the two theoretical structures
- The double bonds ‘resonate’ between the two possible structures
- P-orbitals on each carbon atom overlap forming a delocalised π system
- There are delocalized electrons present above and below the ring
- The delocalisation gives the molecule greater stability
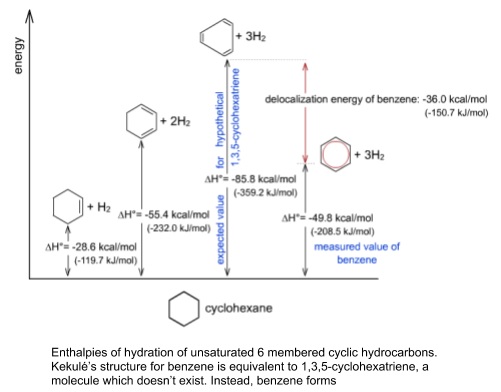
2. Experimental evidence for the structure of benzene
- In Kekulé’s structure the three double bonds would be shorter than the single bonds
- X-ray diffraction showed that all bonds were the same length
- Benzene is much more thermostable than expected from Kekulé’s structure
- Its enthalpy of hydrogenation is much less exothermic than would be expected for three localised double bonds
- This stability is explained by the delocalised π electrons
- Benzene also doesn’t not follow the same patterns of reactivity as cyclohexene
- Undergoes substitution rather than addition reactions e.g. with bromine
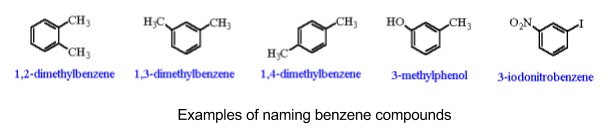
3. Naming aromatic compounds
- Use stem and prefix to generate name
- Stem is always ‘benzene’ unless a more important functional group is present
- Hydrogen atoms on benzene can be replaced in substitution reactions
- Prefix depends on what these substituents are
Substituent |
Prefix |
-Cl |
chloro |
-Br |
bromo |
-NO2 (nitro group) |
nitro |
Alkyl e.g. -C2H5 |
e.g. ethyl |
- For single substituents you don’t need a number
- For two substituents write the group prefixes in alphabetical order
- Number the first group 1-, then the other group relative to it
- For three substituents do the same, always numbering the second and third groups to have the lowest possible numbers
- If there’s more than one of the same group use di- or tri-
- If benzene isn’t the most important functional group it’s not used as the stem
- It acts like an alkyl group instead with the name phenyl e.g. phenylethene
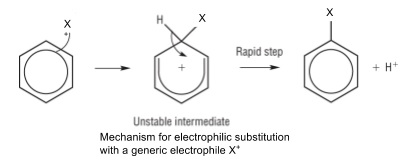
4. Electrophilic substitution
- Benzene is too stable to undergo addition due to the stable delocalised π system
- Undergoes substitution instead
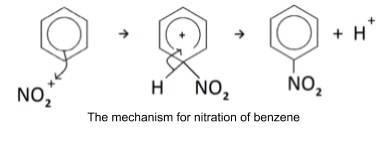
- Nitration: reaction with conc. nitric acid and conc. sulphuric acid catalyst
- Forms nitrobenzene and water
- Overall C6H6 + HNO3 → C6H5NO2 + H2O
- conc. nitric and conc. sulphuric acid mixed in ice bath
- Form nitrating species NO2+
- H2SO4 + HNO3 → NO2+ + HSO4- + H2O
- Benzene added and refluxed at 50oc
- Any higher and further substitution will occur
- conc. nitric and conc. sulphuric acid mixed in ice bath
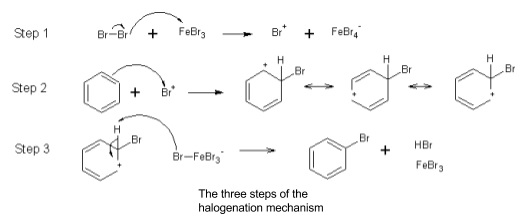
- Halogenation
- Benzene doesn’t react directly with halogens
- Halogen carrier needed - AlX3, FeX3 or Fe
- Electrophile can be represented as X+
- Fe + X2 → FeX- + X+
- FeX3 + X2 → FeX4- + X+
- Halogen carrier is a catalyst, reacts with H+ at the end of the reaction to form HX and regenerate
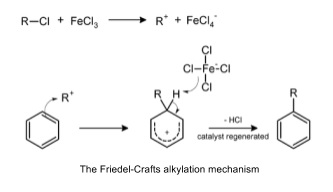
- Friedel-Crafts reaction is electrophilic substitution of an alkyl or acyl group (R+)
- Haloalkane and a halogen carrier e.g. FeX3
- R-X + FeX3 → R+ + FeX4-
- Room temperature
- Multiple substitutions likely because alkyl group donates electrons into the delocalised π system
- Makes it more electrophilic
- Products can be separated by fractional distillation/chromatography
- Acyl chloride can also be used
- Very reactive, but refluxed at 60oc for 30 minutes in anhydrous conditions
- Carbonyl group withdraws electrons from aromatic ring so make it less electrophilic
- Less reactive so only one substitution happens
5. Benzene’s resistance to bromination
- In ethene all electron density is concentrated between the two carbons
- So electron rich and reactive with electrophiles
- In benzene electrons are delocalised around the ring
- So less concentrated between two carbon atoms
- Less able to induce a dipole in halogens to cause them to react
6. Interpreting unfamiliar electrophilic substitutions
- Remember the general mechanism in point 4
- Substitute in the electrophile given in the question for X+
7. Phenols
- Weak acids
- Partially dissociates in water C6H5OH + H2O ⇌ C6H5O- + H3O+
- Acidity shown by reaction with strong bases to form salt + water
- C6H5OH + NaOH → C6H5O-Na+ + H2O
- Doesn’t react with weak bases like carbonates
8. Electrophilic substitution of phenols
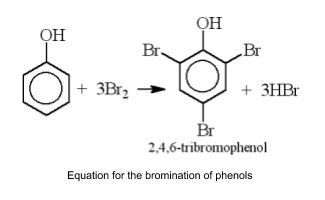
- React with bromine directly to form 2,4,6-tribromophenol (white precipitate)
- Undergoes single substitution with dilute HNO3 at room temperature
- C6H5OH + HNO3 → C6H4(NO2)OH + H2O
- If conc. nitric acid is used get triple substitution to 2,4,6-trinitrophenol
Exam Tip Remember that nitration of phenol doesn’t need conc. nitric acid or a conc. sulphuric acid catalyst like the nitration of benzene does because phenol is more reactive |
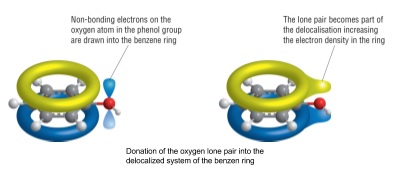
9. Increased reactivity of phenol
- -OH group attached to benzene ring makes ring much more reactive
- Lone pairs of electrons in an oxygen p-orbital overlap with delocalized pi electrons
- This increases the electron density around the ring
- This causes the benzene ring to be more reactive to electrophiles
- This increased reactivity is called activation
10. Directing effects of ring substituents
- OH and NH2 are electron donating groups
- Cause subsitution reactions to happen at 2- and 4- positions relative to them
- Known as the 2- and 4- directing effect
- NO2 is an electron withdrawing group
- Directs substituents to the 3- position
Exam Tip OH, NH2 and NO2 are the only directing groups you need to know, if you’re given any others in the exam you’ll be told whether they’re withdrawing or donating. |
11. Predicting substitution products with the directing effect
- If a benzene ring has a directing substituent and undergoes substitution we can use this to predict where the substitution will occur
- This is useful in synthesis because it makes it easier to get the product we want
- Always remember to think about directing effects if you’re writing a synthesis, otherwise you might get the wrong answer
2. carbonyl compounds
1. Oxidation of aldehydes
2. Nucleophilic addition reactions
- Aldehydes have a C=O as the terminal carbon with a hydrogen atom bonded to it
- Represented by RCHO
- R is a hydrogen atom, alkyl group or benzene ring
- Oxidised by potassium dichromate (K2Cr2O7) and H2SO4 to carboxylic acid
- Ionic equation
- 3RCHO(l) + Cr2O72-(aq) + 8H+ → 2RCOOH + 2Cr3+ + 4H2O(l)
- 3RCHO(l) + Cr2O72-(aq) + 8H+ → 2RCOOH + 2Cr3+ + 4H2O(l)
- Summarised as RCHO + [O] → RCOOH
- Colour change from orange to green
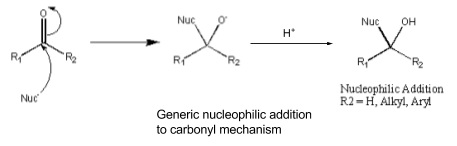
2. Nucleophilic addition reactions
- C=O has a dipole, C is δ+ and O is δ-
- C is an electrophile and attracts nucleophiles
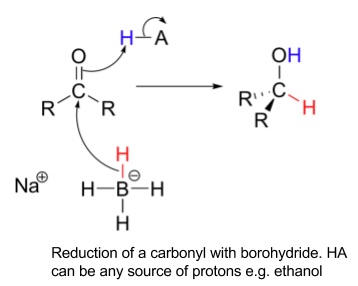
- Sodium tetrahydridoborate(III), NaBH4 (sodium borohydride)
- Na+ and BH4- ion
- BH4- ion is a source of nucleophilic H-, summarised as [H]
- Attacks carbonyl carbon and reduces carbonyl to alcohol
- Reduces aldehyde to primary alcohol and ketone to secondary alcohol
- RCHO + 2[H] → RCH2OH
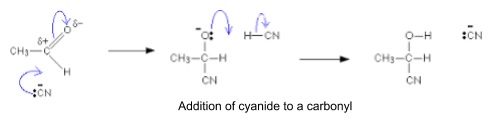
- Hydrogen cyanide, HCN
- Weak acid and dissociates in water
- HCN + H2O → CN- + H3O+
- CN- acts as nucleophile
- Other sources e.g. NaCN
- Reaction needs to be acidified to make carbonyl reactive enough for CN- to attack
- Acid shifts electron density onto O of carbonyl and makes C more positive/reactive
- Also shifts HCN equilibrium to the left so pH 4 is the lowest used to balance these
- Adding CN- allows more carbon atoms to be added to a molecule
- This is hard to do otherwise so this reaction is useful
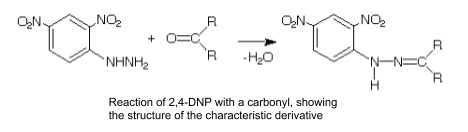
3. 2,4-DNP to test for carbonyls
- Creates yellow/orange precipitate in presence of aldehyde/ketone
- Not with carboxylic acid or ester
- Similar chain length aldehydes/ketones have very similar melting points
- 2,4-DNP derivatives have very different and specific melting points
- Find melting point and compare to table of derivatives to identify aldehyde/ketone
4. Tollen’s reagent to test for carbonyls
- Silver nitrate, nitric acid and ammonia
- Weak oxidising agent
- Silver reduced to give a silver mirror on reaction with an aldehyde
- Ketones can’t be reduced so no reaction
- Ag+ + e- → Ag
- RCHO + [O] → RCOOH
3. carboxylic acids and esters
1. Solubility of carboxylic acids
2. Reactions of carboxylic acids
3. Esters
- -COOH group is very polar and can form hydrogen bonds
- Small carboxylic acids are therefore soluble in water
- In long chain carboxylic acids the non-polar carbon chain is more important so they aren’t soluble in water
2. Reactions of carboxylic acids
- Weak acids so react with bases - metals, metal oxides/hydroxides/carbonates
- Metals
- React with metals above hydrogen in the reactivity series and effervesce H2
- Form salt, stem doesn’t change, suffix becomes -oate
- E.g. ethanoic acid → ethanoate
- 2CH3COOH + 2Na → 2CH3COONa + H2
- Metal oxides
- Form water and metal salt
- CaO(s) + 2CH3COOH(aq) → (CH3COO)2Ca(aq) + H2O(l)
- Group 1 metal hydroxides
- Soluble bases (alkalis) that release OH- into solution
- Neutralise with carboxylic acids to make a salt and water
- KOH(aq) + HCOOH(aq) → HCOOK(aq) + H2O(l)
- Metal carbonates
- React with carboxylic acids to give a salt, water and effervesce CO2
- K2CO3(s) + 2CH3COOH(aq) → 2CH3COOK(aq) + H2O(l) + CO2(g)
- Carbonates can also have only one group 1 atom in them e.g. NaHCO3
- These react exactly the same way
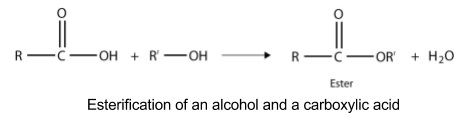
3. Esters
- Contain RCOOR’
- To name:
- The stem is based on the side with the carbonyl group
- The suffix is -oate
- The prefix is based on the side without the carbonyl group
- Two esterification reactions
- Reaction of an alcohol and a carboxylic acid
- Dehydration reaction eliminating water
- Gently heat in the presence of sulphuric acid
- Reaction is reversible and slow so needs to be heated gently
- For large esters reaction needs to be refluxed
- Reaction doesn’t occur with phenols as they’re too unreactive
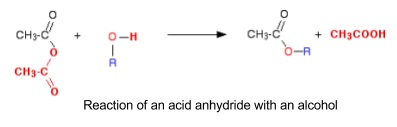
- Reaction of acid anhydrides with alcohols
- Acid anhydrides formed from dehydration of two carboxylic acids
- React with alcohols including phenols to make an ester
- Irreversible so higher yield than carboxylic reaction
- Still slow but can be sped up by gently warming
- Acid anhydrides formed from dehydration of two carboxylic acids
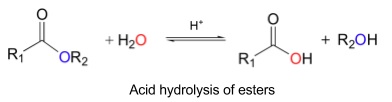
4. Hydrolysis of esters
- Reaction where water breaks down bonds in a decomposition reaction
- Reverse of esterification (dehydration reactions)
- Happens in hot aqueous acid to form carboxylic acids and alcohols
- Needs to be refluxed
- Reversible
- Needs to be refluxed
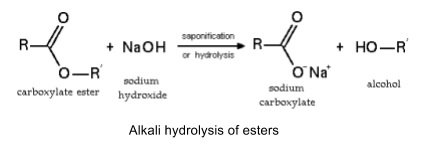
- Also happens in hot aqueous alkali to form carboxylic salts and alcohols
- Irreversible
- Used to make soap (saponification)
- Irreversible
5. Formation of acyl chlorides
6. Uses in synthesis
- Acyl chlorides are very reactive and contain R-COCl
- To name:
- Use longest carbon stem as stem
- Suffix is -oyl chloride
- e.g. ethanoyl chloride
- Use longest carbon stem as stem
- Form by replacing -OH group in carboxylic acid with Cl
- One way is using SOCl2
- Liquid at room temperature so just mix with acid
- CH3COOH + SOCl2 → CH3COCl + SO2 + HCl
- Vigorous reaction and produces HCl gas so difficult to do
- Liquid at room temperature so just mix with acid
6. Uses in synthesis
- Acyl chlorides are a very useful group in making a range of compounds
- Esters
- Acyl chlorides react with alcohols including phenols to give esters
- So can use it to make an ester from a phenol
- CH3COCl + CH3CH2CH2OH → CH3COOCH2CH2CH3
- Irreversible and higher yield than reaction with carboxylic acids
- Vigorous and produces HCl fumes so harder to do
- Acyl chlorides react with alcohols including phenols to give esters
- Carboxylic acids
- Small acid chlorides hydrolise quickly with water to form the acid
- CH3COCl + H2O → CH3COOH + HCl
- Very exothermic and HCl produced so difficult
- Small acid chlorides hydrolise quickly with water to form the acid
- Primary amides (nitrogen atom bonded to carbon and two hydrogens)
- Reaction of an acyl chloride with ammonia produces a primary amide
- RCOCl + 2NH3 → RCONH2 + NH4Cl
- Reaction of an acyl chloride with ammonia produces a primary amide
- Naming primary amides
- Stem is longest carbon chain
- Suffix is -amide e.g. ethanamide for CH3CH2CONH2
- Stem is longest carbon chain
- Secondary amides (nitrogen atom bonded to carbon and one hydrogen)
- Acyl chloride reacts with a primary amide to make a secondary amide
- Often called an N-substituted amide
- Acyl chloride reacts with a primary amide to make a secondary amide