A2-LEVEL OCR ChEMISTRY NOTES
TOPIC 3: analysis
1. Chromatography and quantitive analysis
1. Interpreting TLC chromatograms
2. Interpreting GC chromatographs
3. Qualitative analysis of organic compounds
- Stationary phase - thin piece of inert material e.g. glass with an absorbent chemical e.g. aluminium oxide
- Mobile phase - organic solvent which moves vertically up the TLC plate
- Samples are spotted onto the TLC plate and it’s put into a tank with solvent at the bottom
- Separates components of the mixture due to their different affinities for the absorbent chemical on the TLC plate
- Rf value is a way of quantifying afinities for the stationary phase
- Rf = distance moved by componentdistance moved by solvent front
- Rf values for different compounds in specific stationary/mobile phases are tabulated, so the identity of each component can be worked out
2. Interpreting GC chromatographs
- Stationary phase - liquid or solid coating the inside of the coiled GC tube
- Mobile phase - inert carrier gas e.g. helium or nitrogen
- Volatile mixtures are separated as they’re carried through the GC tube due to their different affinities for the stationary phase
- Retention time is the equivalent of Rf values for GC
- Length of time for a compound to pass through the GC
- Plotted as a graph of adsorption in the detector in the GC against time
- Compounds with high affinity for the stationary phase take longer to come through
- There are also tables for retention times of different compounds with different stationary so identities can be worked out just from retention times
- However, GC is often paired with mass spectrometry to give further analysis of each component as it comes out of the GC
- Concentrations are proportional to the area under peaks on an absorption/time graph
- So relative concentrations can be found from peak areas without further analysis
- External calibration curves need to made to determine absolute concentrations
- A known amount of a pure compound is passed through the GC and it’s peak area and retention times found
- The peak area for the known compound can then be compared to unknown peaks to work out their concentrations
3. Qualitative analysis of organic compounds
- May need to perform more than one of the tests to determine all the functional groups in a compound
Functional group |
Reagents |
Observations |
Unsaturation (alkenes) |
Bromine water |
Bromine water decolourises when added to an unsaturated compound |
Haloalkanes |
Aqueous silver nitrate and ethanol |
If a precipitate forms a halogen is present White precipitate - chloroalkane Cream precipitate - bromoalkane Yellow precipitate - iodoalkane |
Carbonyl |
Brady’s reagent (2,4-DNP) |
Yellow or orange precipitate forms if ketone/aldehyde present |
Aldehydes |
Fehling’s solution Tollen’s reagent Acidified potassium dichromate |
Fehling’s solution - blue solution forms dark red precipitate Tollen’s reagent forms a silver mirror Orange acidified potassium dichromate solution turns green |
Aliphatic carboxylic acids |
Universal indicator/pH probe Reactive metal e.g. magnesium Metal carbonate e.g. calcium carbonate |
pH will be that of a weak acid Metal and carbonate will effervesce with hydrogen and CO2 respectively |
Phenols |
Universal indicator/pH probe No reaction with CO32- |
pH will be that of a weak acid No effervescence with a carbonate |
Alcohols |
Acidified potassium dichromate |
Primary - colour change from orange to green as alcohol is oxidised to carbonyl then carboxylic acid Secondary - colour change as alcohol oxidised to ketone Tertiary - no colour change |
Exam Tip Remember that qualitative analysis is only based on an observable change and doesn’t provide information on the actual quantities of a compound that might be present |
2. spectroscopy
1. NMR
- Non destructive technique
- Analyses radio wave energy required to flip spins of certain isotopes (1H/13C) in a magnetic field
- Energy required depends on chemical environment so can be used to work out the different kinds of isotope atoms in a molecule and how many there are
- The fewer electrons there are around the nucleus being investigated the higher its chemical shift will be
- Chemical shift, δ, is how the energy required to flip the spin is measured
- Its units are ppm and shifts are measured relative to an internal standard (TMS)
- 1.1% of carbon is 13C which unlike 12C has spin so can be used in NMR
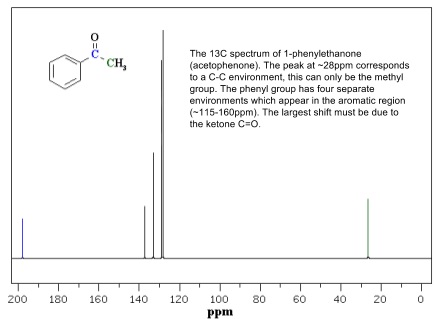
- Each environment present in the molecule gives one peak, so the number of peaks is equivalent to the number of environments
- More than one carbon can be in each environment (equivalents) so number of peaks isn’t the same as total number of carbons
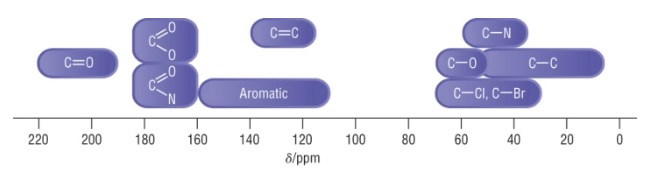
- Shifts for carbon atoms in different environments range from 0-220 ppm
- Use these shifts to assign the environments of peaks in a spectrum
- Consider the number of environments and possible functional groups when assigning spectra
- Combine the information about the functional groups to predict a plausible molecule containing them all
- TMS is used as a reference standard as it’s carbon has a very low shift, so most carbon environments appear above it
Exam Tip You’re given a table of relevant chemical shifts for 1H and 13C NMR in the databook, you don’t need to memorise the values |
3. 1H NMR
- Each chemical environment gives a different chemical shift so number of peaks is equivalent to number of environments
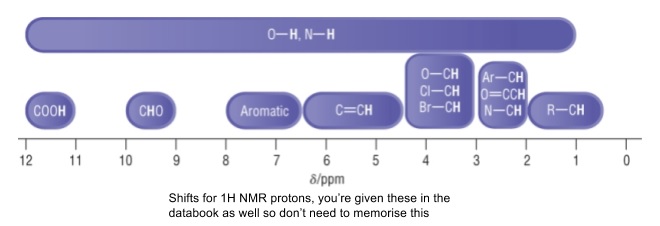
- Shifts are from 0-12 ppm
- Area under each peak is the ratio of protons in each environment
- This is given as an integration trace on the spectrum
- TMS is used as a standard as it has 12 equivalent protons which give a single intense peak that can be assigned as 0 ppm
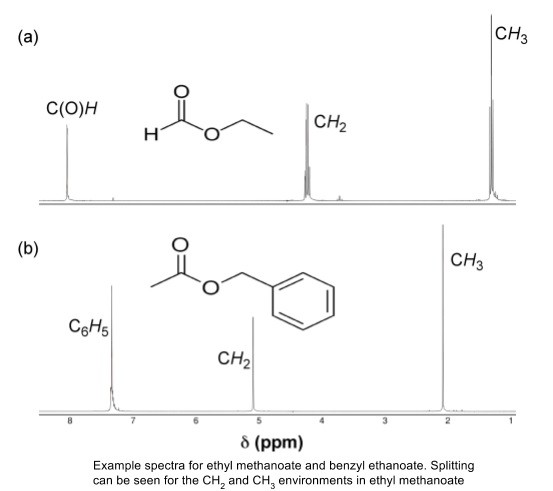
- Protons on adjacent carbons cause the signal for an environment to be split into multiple peaks
- n + 1 rule: number of peaks in splitting pattern = number of protons on adjacent carbons + 1
- Singlet means no protons on adjacent carbons, doublet means one proton etc
Exam Tip The triplet-quartet pairing is very common and normally implies that there’s a CH2CH3 group present |
- Triplets and quartets can be caused by two or three protons on different neighbouring atoms, they don’t always have to be CH2 or CH3 groups
4. Predicting NMR spectra from molecules
- Identify all the different environments
- Put a peak for each environment in the right region of the spectra depending on what functional group it’s part of
- If it’s a 1H-NMR spectrum add the number of protons to each environment
- Also include any splittings that might be present
- NMR spectra are normally made for compounds in solution
- Organic solvents are needed for organic compounds but these contain lots of proton environments that would confuse the spectra
- 1H is the only isotope of hydrogen with spin
- Deuterium (2H) doesn’t have spin so it is substituted for 1H in common solvents
- CDCl3 is commonly used
- Chemical environments have certain chemical shifts in spectra
- These are measured relative to an internal standard, normally TMS
- This gives one strong peak at a shift normally lower than all other environments in 13C and 1H NMR which can be assigned as 0 ppm
- TMS has a low boiling point and is unreactive so won’t affect the compound and can be boiled off at then end to recover the sample
- It can be difficult to identify OH/NH protons in NMR
- Their peaks are normally broad,
- Show no splitting
- Come at a range of shifts depending on concentration and solvent
- OH/NH protons exchange rapidly with other OH/NH protons in the sample
- If the sample is vigorously shaken with D2O the protons will be exchanged for deuterium atoms
- These aren’t spin active, so don’t show up in spectra
- This means peaks due to OH/NH protons disappear from a spectra when shaken with D2O
- OH/NH protons also don’t generally cause splitting of nearby protons due to the fact that they are exchanged so frequently
- Often more than one analytical technique is combined to fully work out the structure and properties of a compound
- Elemental analysis gives the relative proportions of elements in a compound
- This can be used to find the empirical formula
- Mass spectra can be used to find the molecular mass, which can be combined with the empirical formula to find the molecular formula
- Fragmentation patterns also give clues about structure
- Isomers of the same compound will have the same molecular mass and empirical formula, so other techniques may be needed to distinguish between them
- IR spectroscopy gives information about the bonds and likely functional groups and is non-destructive
- Members of the same homologous series will have similar spectra
- NMR spectroscopy gives information about the number and types of environments in a molecule
- Proton NMR also gives information about the number of protons in each environment
- To find the identity of a molecule given a range of analytical techniques
- Identify the empirical and molecular formulas from MS and elemental analysis
- Use IR to find the nature of the bonds and functional groups
- Use NMR to find the number and type of environments