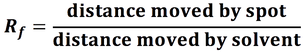A-LEVEL AQA ChEMISTRY NOTES
chromatography
Chromatography
- Chromatography is a separation technique used for separating and identifying the species present in a mixture.
- The mobile phase is the substance in chromatography that carries the soluble components of the mixture
- The stationary phase is the substance in chromatography that holds back the components that are attracted to it.
- Each component in the mixture has a different level of solubility in the mobile phase and retention by the stationary phase, resulting in the separation of these components
Thin Layer Chromatography
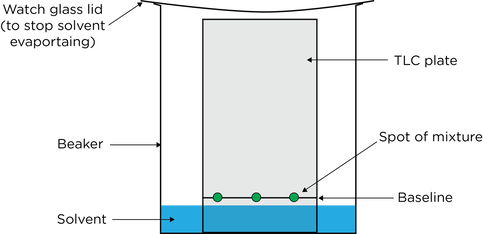
- In thin layer chromatography a plate is coated with a solid and a solvent moves up the plate
- An organic solvent is used as the mobile phase
- A sheet coated with a thin layer of silica gel or alumina acts as the stationary phase
- Thin layer chromatography can be used to separate and identify amino acids by their Rf values.
- Different amino acids have different R groups, so they all have different affinities for the solvent. Therefore, moving up the plate at different rates depending on their affinity.
- Amino acids are colourless, so to make them visible a developing agent is used, such as ninhydrin or ultraviolet light
- Rf values are affected by temperature, a spot of a pure substance is often added to the baseline for comparison. If the correct conditions are used the Rf values can be compared to database values
Column Chromatography
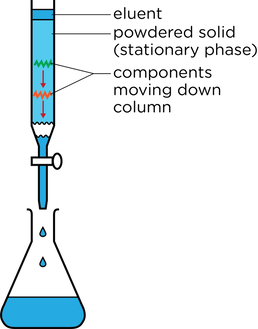
- In column chromatography (CC) a column is packed with a solid and a solvent moves down the column
- Silica or aluminium oxide is packed into a narrow column and acts as the stationary phase
- The mixture is dissolved in a solvent which is poured through the column. This acts as the mobile phase
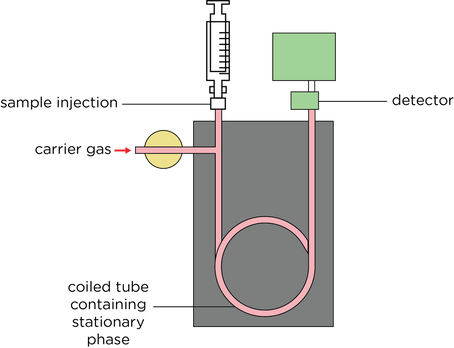
Gas Chromatography
- In gas chromatography (GC) a column is packed with a solid or with a solid coated by a liquid, and a gas is passed through the column under pressure at high temperature.
- Stationary phase is a solid or liquid coating inside a long-coiled tube
- An inert gas such as N2 acts as the mobile phase
- The time taken for a component to leave the coil is called the retention time. This can be compared with standards to identify different substances.
- A chromatograph shows these retention times as a series of peaks with the area under each peak being proportional to the amount of that component present
- Mass spectrometry can be used to analyse the components separated by GC
- The mass spectrum of each component can be compared to spectra in a database, allowing the components to be identified with greater certainty.