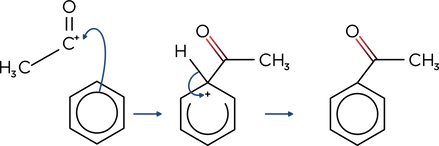A-LEVEL AQA ChEMISTRY NOTES
aromatic chemistry
Bonding in Benzene
- Compounds that contain a benzene ring are aromatic.
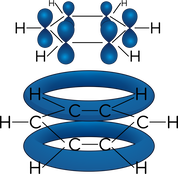
- Benzene has a planar cyclic structure consisting of a ring of carbon atoms, each with a single hydrogen atom attached that sticks out into a flat plane
- Benzene has a delocalised electron system, arising due to the overlap of one p-orbital from each carbon atom, above and below the plane of the ring.
- Electrons are shared evenly, so all bond lengths are the same, between that of a single bond and a double bond
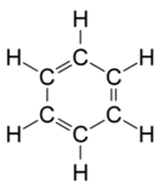
- The delocalisation of electrons gives benzene extra stability than the theoretical cyclohexa-1,3,5-triene molecule, which has alternating double and single bonds. This is reflected by the hydrogenation of benzene being less exothermic than predicated by cyclohexa-1,3,5-triene molecule.
- Benzene undergoes electrophilic substitution reactions rather than electrophilic additions because a lot of energy is needed to disrupt the delocalised system
Electrophilic Substitution of Benzene
- The benzene ring is a region of high electron density, which means it attracts electrophiles
- Nitration of benzene: C6H6 + HNO3 → C6H5NO2 + H2O
- Generation of the electrophile:
- HNO3 + H2SO4 → H2NO3^+ + HSO4^- → H2O + NO2^+ + HSO4^-
- Generation of the electrophile:
- Regeneration of sulphuric acid catalyst: H+ + HSO4- → H2SO4
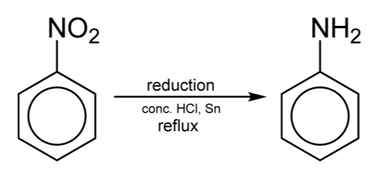
- Nitrated benzene rings can be used in the production of explosives, dyes and phenylamine.
- Friedel-Crafts acylation reactions involve adding an acyl group to benzene. e.g.
- CH3COCl + AlCl3 → CH3CO+ + AlCl4-
- A halogen carrier, such as AlCl3, is used to convert the acyl chloride into an electrophile