A-LEVEL BIOLOGY OCR NOTES
BIOLOGICAL MOLECULES
Water
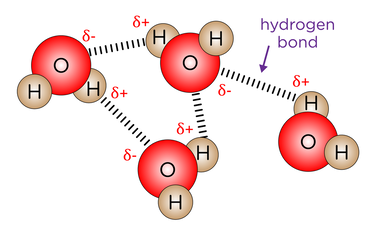
- Water molecules consist of 2 hydrogen molecules covalently to an oxygen molecule.
- The molecules are slightly polar because the oxygen nucleus pulls the shared electrons away from the hydrogen nuclei. Giving the oxygen nuclei a δ- charge, and the hydrogen nuclei a δ+ charge.
- The polarity of water causes attraction between water molecules. This force of attraction is called a hydrogen bond.
Property of Water |
Why is it useful |
Liquid medium |
Provides habitats for aquatic organisms, medium for chemical reactions & used for transport |
Important metabolite |
Used in hydrolysis & condensation reactions |
High specific heat capacity |
Keeps aquatic & cellular environments stable |
High latent heat of vaporisation |
Evaporation has a cooling effect on organisms |
Cohesion of molecules |
Water is drawn up the xylem |
Surface tension |
Allows pond-skaters to walk on the surface |
Good solvent and transport medium |
Dissolves ionic and polar molecules, allowing them to easily be transported |
Good reaction medium |
The cytoplasm in cells is an aqueous solution where many chemical reactions happen |
Incompressible |
Can prevent plants from wilting & act as a hydrostatic skeleton for invertebrates |
Monomers & Polymers
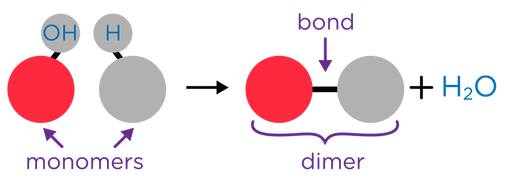
- Monomers are individual molecules that make up a polymer.
- Polymers are long chains that are composed of many individual monomers that have been bonded together in a repeating pattern.
- Condensation Reactions occurs when two molecules combine to form a more complex molecule with the removal of water.
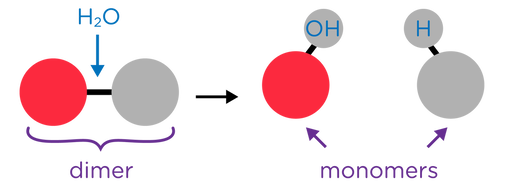
- Hydrolysis Reactions occurs when larger molecules are broken down into smaller molecules with the addition of water.
Carbohydrates
- Monosaccharides are the simplest carbohydrates, consisting of only one sugar molecule (e.g. Glucose, Fructose & Galactose)
- Ribose sugars (pentose) are found in many important biological molecules such as ribonucleic acid (RNA), ATP, NAD
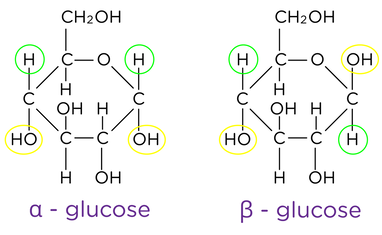
- Glucose is a hexose sugar with 2 isomers
- Disaccharides are sugars that are composed of two monosaccharides joined together in a condensation reaction, forming a glycosidic bond.
Disaccharide |
Constituent monosaccharides |
Maltose |
2 × a-glucose |
Sucrose |
a-glucose and fructose |
Lactose |
b-glucose and galactose |
- Polysaccharides are formed by many monosaccharides joined together.
- Amylose, amylopectin (starch) is the main polysaccharide energy store in plants, is composed of α-glucose.
- In animals, the polysaccharide energy store is called glycogen, composed of α-glucose.
- Cellulose is a structural component of plant cell walls, composed of long unbranched chains of b-glucose.
Lipids
- Fatty acids can be:
- Saturated – there are no C=C bonds and the molecule has as many hydrogen atoms as possible.
- Unsaturated – there is at least one C=C bond, therefore the molecule contains fewer hydrogen atoms than is maximally possible.
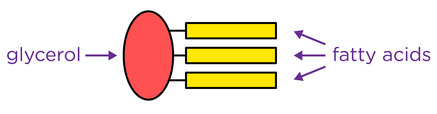
- A triglyceride molecule is formed by joining one molecule of glycerol to three fatty acids through three condensation reactions, forming ester bonds.
- Triglycerides have key roles in respiration and energy storage due to its insolubility and high carbon to hydrogen ratio.
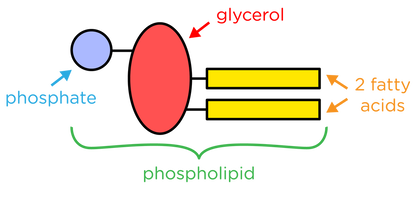
- Phospholipids replace one of the fatty acid chains in triglycerides with a phosphate molecule.
- The hydrophobic tails and hydrophilic heads of phospholipids allow them to form phospholipid bilayers
Proteins
- Amino acids are the monomer units used to make proteins.
- The 20 naturally occurring amino acids only differ in their R groups
- Dipeptides are formed when two amino acids are joined together by a condensation reaction, forming a peptide bond.
- A polypeptide is a polymer made of many amino acids joined together by peptide bonds.
- A protein may contain one or more polypeptide chains.
- There are four structural levels:
Level |
Definition |
Bond Type |
Primary |
The specific sequence of amino acids in a polypeptide chain |
Peptide Bonds |
Secondary |
The curling or folding of the polypeptide chain into α-helices and β-pleated sheets due to the formation of hydrogen bonds |
Hydrogen Bonds |
Tertiary |
The overall specific 3-D shape of a protein, which is determined by interactions between R groups and the properties of R groups |
Hydrogen bonds Ionic bonds Disulphide bridges |
Quaternary |
The specific 3-D shape of a protein that is determined by the multiple polypeptide chains and/or prosthetic groups bonded together |
Hydrogen bonds Ionic bonds Disulphide bridges |
Inorganic Ions
- Inorganic ions are atoms or molecules with an electric charge, containing no carbon.
- Cations are positively charged ions
- Anions are negatively charged ions
- Inorganic ions occur in solution in the cytoplasm and body fluids of organisms, some in high concentrations and others in very low concentrations
- Each type of ion has a specific role, depending on its properties
- Hydrogen ions determine the pH of bodily fluids. The higher the concentration, the lower the pH
- Sodium ions are used in the co-transport of glucose and amino acids across cell membranes and transmission of nervous impulses
- Phosphate ions are essential components of DNA, RNA, nucleotides & ATP
- Calcium ions regulate protein channels, impulse transmission and harden body parts like teeth.
- Potassium ions play a role in muscle contraction, nervous transmission. active transport and maintaining turgidity in plant cells.
- Ammonium ions and Nitrate ions are part of the nitrogen cycle and a source of nitrogen for biological molecules
- Hydrogencarbonate ions are formed when carbon dioxide dissolves in blood. They are important in the transport of carbon dioxide in the blood and the regulation of blood pH
CO2 + H2O ⇌ HCO3^- + H^+
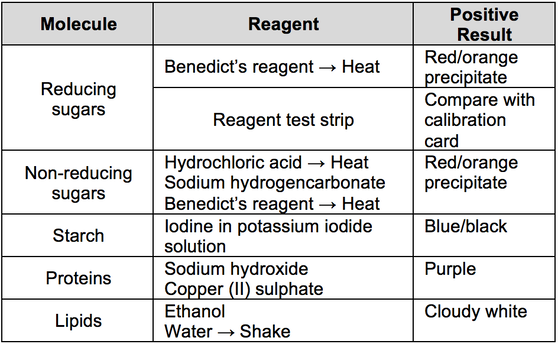
Biochemical Tests
Globular Proteins
- A globular protein is protein with a spherical shape that is soluble in water; they typically have metabolic roles
- Globular proteins have a few important properties:
- They are roughly spherical in shape, with hydrophobic R groups on the inside and hydrophilic R groups on the outside. They are therefore soluble in water
- They have very specific shapes; this allows them to carry out very specific functions
- Haemoglobin is a conjugated globular protein, made of 4 polypeptide chains and 4 haem prosthetic groups which contain Fe2+.
- Insulin is composed of two polypeptide chains, joined together by disulphide links. The specificity of the shape allows binding to cell membrane receptors.
- Pepsin has hydrogen bonds, disulphide link and few basic R groups to keep it functional in low pH stomach acid.
Fibrous Proteins
- Fibrous proteins have some similar properties to each other:
- They contain long polypeptide chains with repeating sequences of amino acids
- The amino acids have non-polar R groups, so the proteins are insoluble in water
- The polypeptide chains are able to form fibres which make the proteins stronger
- Collagen is used to make bones, tendons and are found in artery walls to help resist the high pressure
- Keratin contains high amounts of cysteine, resulting in disulphide links forming between the two polypeptide chains, which makes the molecule very hard and strong. It is found in fingernails, hooves and horns.
- Elastin has the ability to stretch and recoil. Elastin is stretchy due to coiling of the elastin molecules and cross-links that keep the molecules together. It is found in the lungs, bladder and blood vessel walls.